SNAS and the Brain: Brain Fog, Brain Health, and Mental Health in Systemic Nickel Allergy
If you have systemic nickel allergy syndrome (SNAS) and brain fog, you are not imagining it. There are real, documented biological mechanisms connecting the nickel you eat to the way your brain feels. Let's walk through what the research actually says — and what you can do about it.
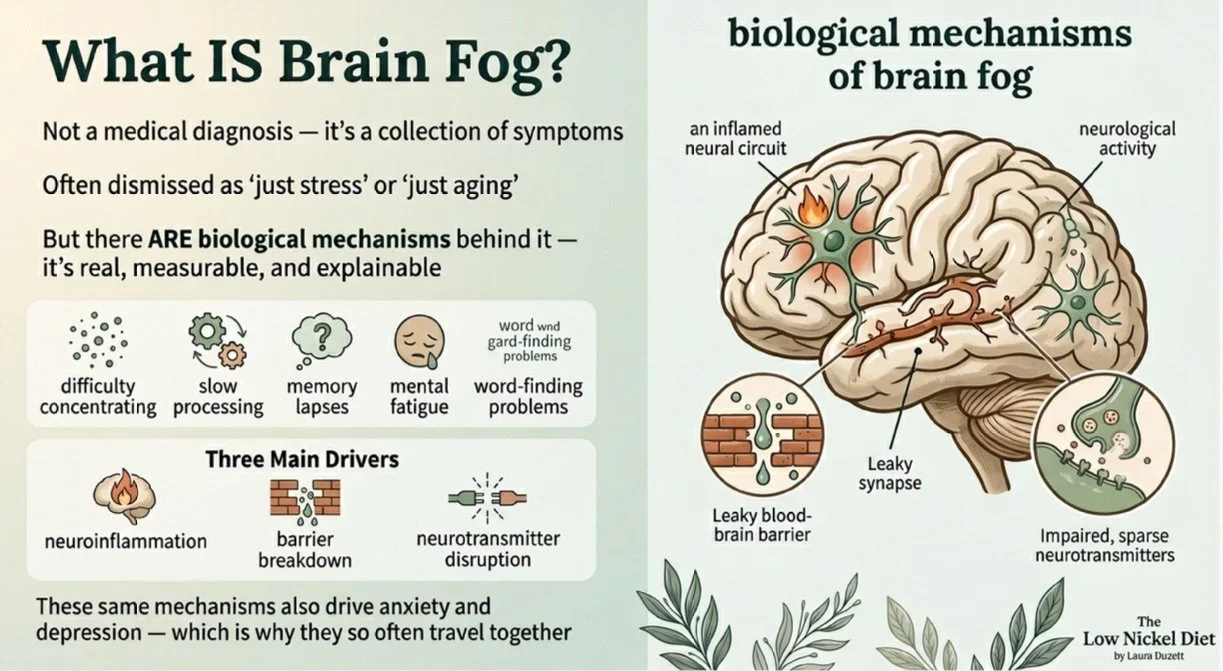
What Brain Fog Actually Is (and Isn't)
Brain fog is not a medical diagnosis. It is a collection of symptoms — difficulty concentrating, memory lapses, mental fatigue, slow processing, word-finding problems — that share real biological mechanisms. For people with SNAS, these symptoms frequently accompany the more recognized skin and GI manifestations, and they are often the most distressing ones.
The problem is that brain fog gets dismissed. It gets chalked up to stress, poor sleep, or "just getting older." But when you understand the pathways that connect nickel ingestion to neuroinflammation, it stops being mysterious and starts being mechanistically predictable.
Here is what is happening under the surface.
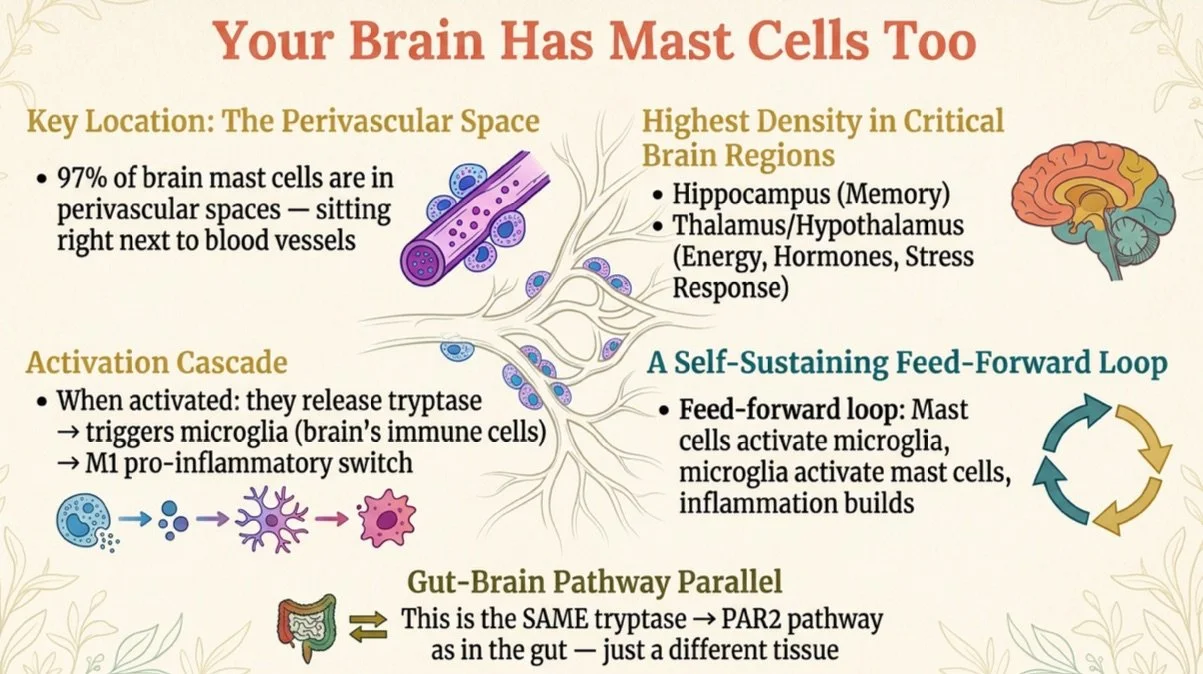
Your Brain Has Mast Cells — and SNAS Activates Them
You probably associate mast cells with skin reactions or gut symptoms. But your brain has its own population of resident mast cells, and approximately 97% of them sit in perivascular spaces — right next to blood vessels, astrocytes, and microglia (Dong et al., 2014; PMID: 25436584).
The highest concentrations are in the hippocampus (memory and learning), the thalamus and hypothalamus (energy regulation and the stress response), and around the third ventricle (which senses systemic inflammation even without a blood-brain barrier breach).
When brain mast cells degranulate, they release tryptase that activates PAR2 receptors on microglia — the brain's resident immune cells. This flips microglia into their pro-inflammatory M1 state, and those activated microglia release TNF-alpha, IL-6, and reactive oxygen species that feed right back to the mast cells (Zhang et al., 2016; PMID: 27050634). The result is a feed-forward loop of chronic, low-grade neuroinflammation.
That loop is brain fog.
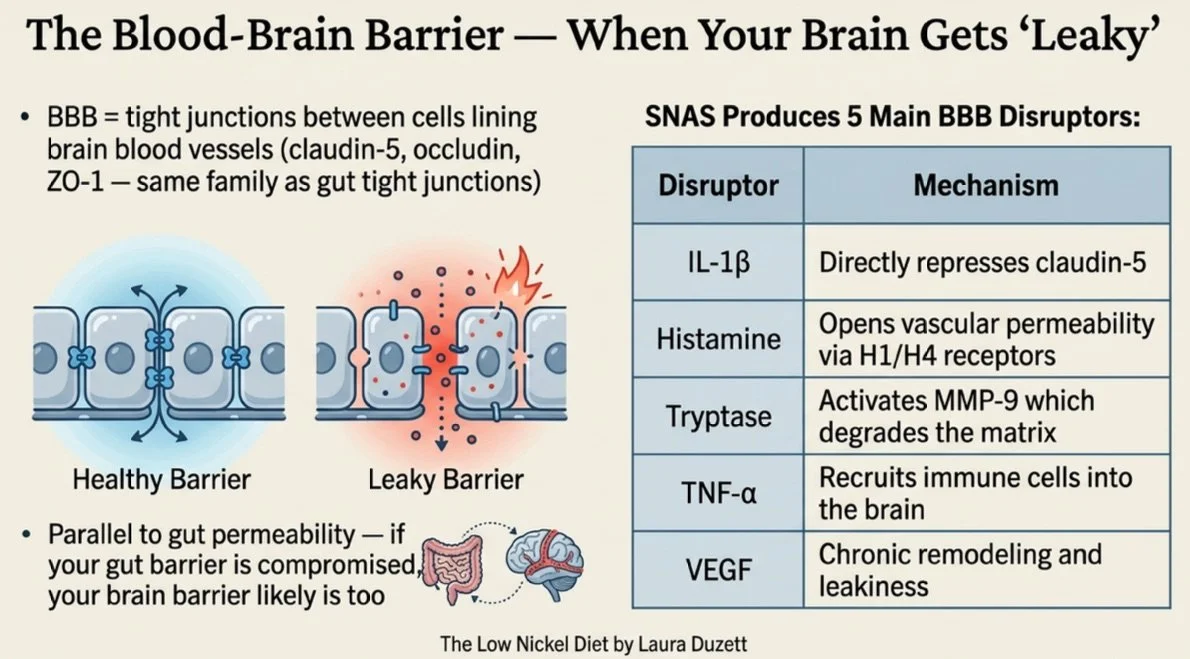
Five Ways SNAS Breaks Down Your Blood-Brain Barrier
The blood-brain barrier (BBB) is made of tight junction proteins — claudin-5, occludin, and ZO-1 — that keep harmful molecules out of your brain tissue. SNAS produces at least five mediators that compromise these junctions (Galea, 2021; PMID: 34497379):
When the BBB becomes permeable, inflammatory molecules and immune cells that should stay in the bloodstream gain access to brain tissue. This compounds the mast cell-microglia loop already happening inside the brain.
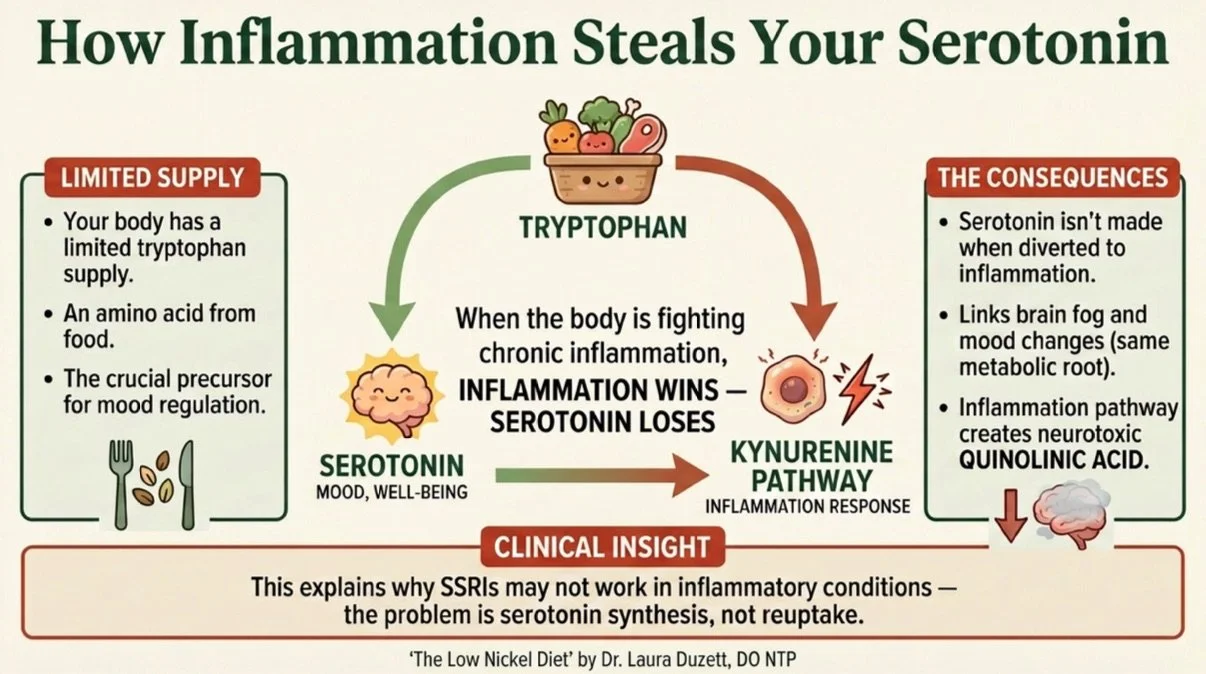
The Kynurenine Pathway: How Inflammation Steals Your Serotonin
Here is something that may reframe how you think about mood changes in SNAS.
Between 90 and 95 percent of dietary tryptophan is metabolized through the kynurenine pathway — not the serotonin pathway. Under normal conditions, this is fine. But under chronic inflammation, pro-inflammatory cytokines like IFN-gamma and TNF-alpha induce the enzyme IDO, which shunts even more tryptophan away from serotonin production and into the kynurenine pathway (Savitz, 2020; PMID: 30980044).
The kynurenine pathway then forks: astrocytes produce KYNA (neuroprotective), but activated microglia produce QUIN (neurotoxic — an NMDA receptor agonist that causes excitotoxicity and neuronal death).
This is why brain fog and mood changes so often go together in SNAS. They share the same metabolic root. It is also why SSRIs may not fully address mood symptoms in inflammatory conditions — the problem is not always reuptake. Sometimes the problem is that serotonin simply is not being produced in adequate amounts.
Nickel as an Endocrine Disruptor: The GH/IGF-1 Connection
A landmark study from Sapienza University (n=1,128) found that nickel allergy is significantly associated with impaired growth hormone (GH) and IGF-1 secretion (Krysiak et al., 2021; PMID: 33419306). The mechanism: nickel ions compete with calcium at pituitary voltage-gated channels, blocking GH vesicle release. MRI findings in the study showed higher prevalence of Empty Sella syndrome.
Why does this matter for your brain? IGF-1 is directly neuroprotective. It supports neuronal survival, synaptic plasticity, and new brain cell growth in the hippocampus. Low IGF-1 also impairs frontolimbic circuit plasticity, which compounds with the kynurenine pathway disruption for a dual hit on mood.
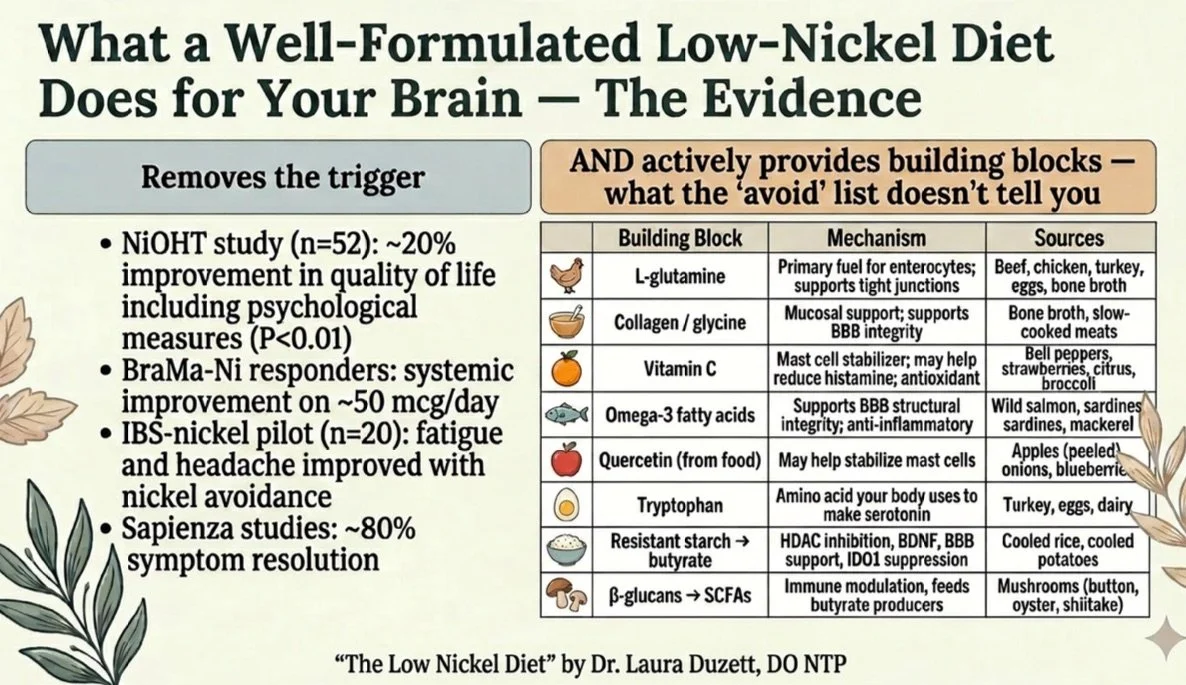
The good news: low-nickel diet studies show approximately 80 percent symptom resolution across multiple Sapienza cohorts, suggesting that removing the nickel trigger may allow GH/IGF-1 function to recover.
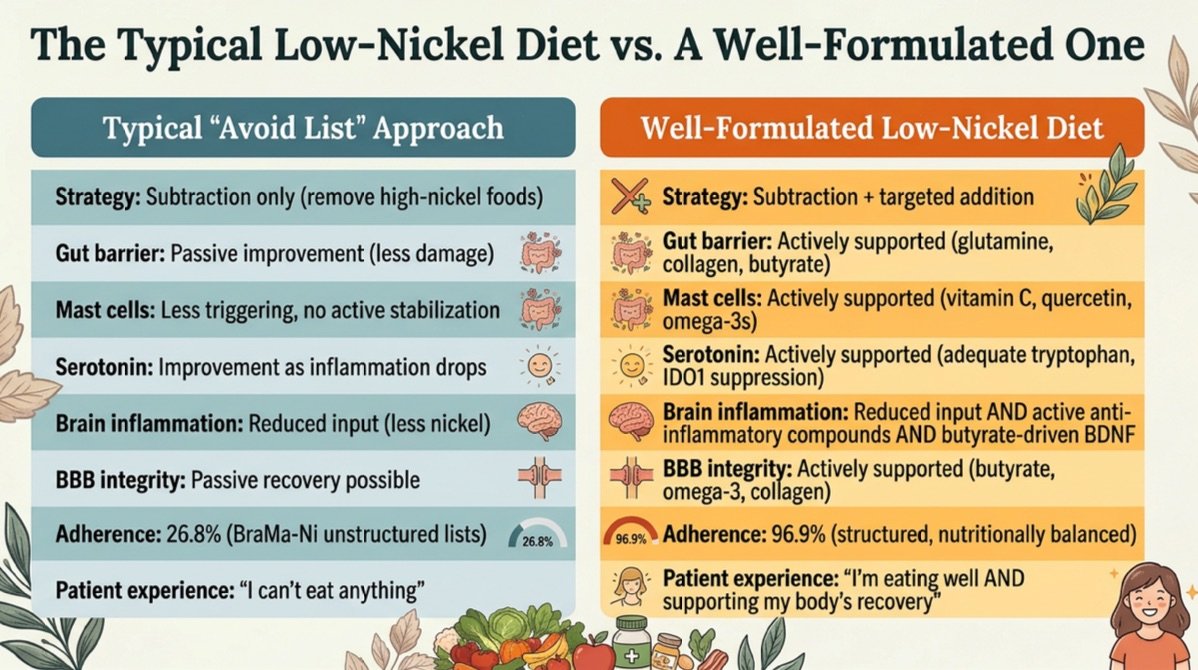
The Well-Formulated Low-Nickel Diet: Subtraction Plus Targeted Addition
A well-formulated low-nickel diet is not just an avoidance list. It is a subtraction-plus-targeted-addition strategy that actively supports the body's natural ability to heal while removing the nickel trigger.
Unstructured avoidance lists achieve only 26.8 percent adherence (Braga et al., 2013; PMID: 24067467). A structured, nutritionally balanced protocol achieves 96.9 percent. The difference matters — especially for your brain.
When you reduce nickel intake to approximately 50 mcg/day, the plate naturally shifts toward animal proteins. This shift provides building blocks your brain needs: L-glutamine for gut barrier integrity, tryptophan for serotonin production, omega-3 fatty acids for BBB structural support, and vitamin C as a mast cell stabilizer. The Low Nickel Diet cookbook makes this practical.
But the real star for brain health may be what happens in your gut.
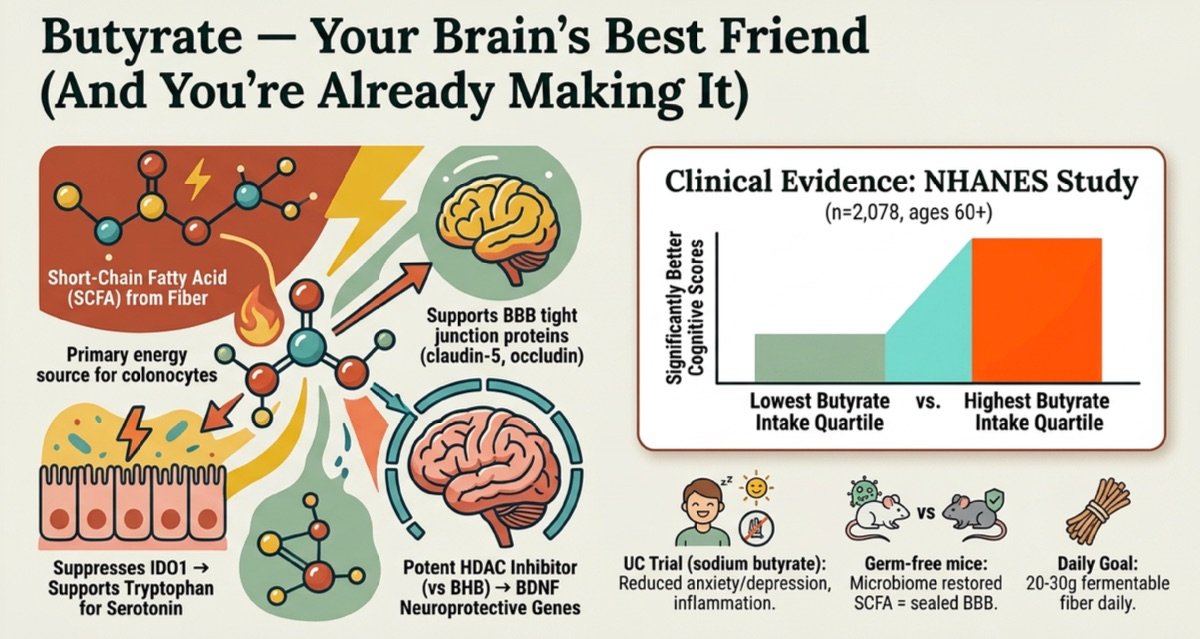
Butyrate from Resistant Starch: Your Brain's Underrated Ally
Butyrate — a short-chain fatty acid produced when gut bacteria ferment fiber — is actually a more potent HDAC inhibitor than BHB, the ketone body (Shimazu et al., 2013; PMID: 23223453). It upregulates BDNF (brain-derived neurotrophic factor), suppresses IDO1 (preserving serotonin), and supports BBB tight junction proteins.
Population-level data backs this up: in the NHANES cohort (n=2,078, ages 60+), the highest butyrate intake quartile showed significantly better cognitive scores. And in a 12-week RCT (n=36), 600 mg/day of sodium butyrate reduced both anxiety/depression scores and inflammatory markers.
The practical question: how do you produce butyrate on a low-nickel diet?
Resistant starch largely survives reheating, so cooled rice and potatoes can be warmed and still provide butyrate substrate.
The Ketone Option: When to Consider It
For severe or refractory brain fog, a low-nickel ketogenic approach or exogenous ketone supplementation may provide additional support. BHB directly inhibits the NLRP3 inflammasome and activates GPR109A on microglia (where it is actually stronger than butyrate). The BENEFIC trial (n=122) showed improved memory and a 230 percent increase in brain ketone uptake with 30g/day MCT oil over six months (Fortier et al., 2021; PMID: 33103819).
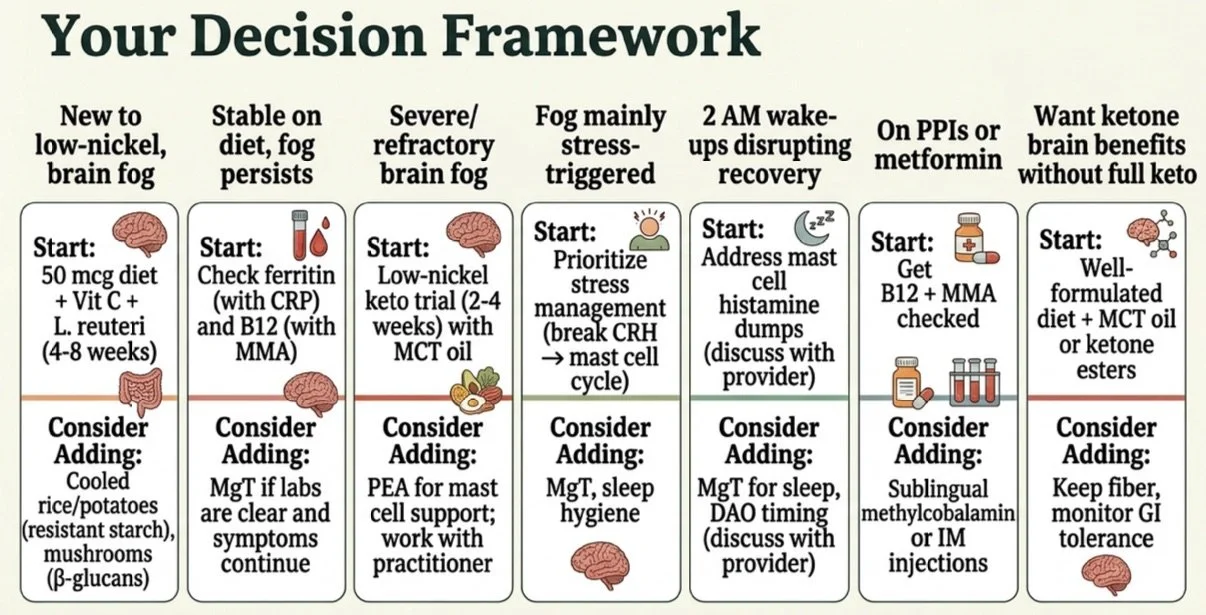
Practical Steps: What to Do and What to Ask Your Doctor
Your Decision Framework
Labs to Discuss with Your Doctor
Your provider can run these to help rule out nutritional deficiencies that mimic or compound brain fog:
Ferritin + CRP: Cognitive threshold is approximately 50 ng/mL (or 100+ with active inflammation). CRP reveals whether ferritin is falsely elevated by inflammation.
B12 + MMA (methylmalonic acid): Subclinical B12 deficiency can begin below 350 pg/mL — well above most lab "deficient" cutoffs. MMA confirms tissue-level deficiency.
IGF-1: If low, consider requesting an endocrinology referral — nickel may be suppressing pituitary GH secretion.
If you are not sure whether SNAS testing applies to your situation, start there.
For more on supplements that may support SNAS management, remember the golden rule: only after your foundational diet is in place, one supplement at a time, and ideally with practitioner guidance.
The Gut-Brain Connection Is Real — and You Can Work With It
The gut-brain axis is not a metaphor. Your vagus nerve carries signals directly from your inflamed gut to your brainstem, hypothalamus, and amygdala. When your gut mast cells degranulate in response to nickel, your brain gets the message in real time — and responds with fatigue, social withdrawal, and reduced cognitive flexibility. This is called sickness behavior, and in chronic SNAS, it can become the baseline rather than the exception.
The flip side is equally real. When you reduce the nickel trigger and support butyrate production, you are providing your body with the building blocks to restore barrier integrity, calm neuroinflammation, and preserve serotonin production.
You did not cause this. But you can work with it.
Watch the Full Deep Dive
This article covers the highlights, but there is much more in the full presentation — including the HPA axis stress cycle, sleep and glymphatic clearance, and a deeper walk through the supplement evidence.
Download the Nourish Forward Symptom Tracker — a free tool to track your brain fog, energy, and other symptoms alongside your diet changes. Because what gets measured gets managed.
This article is for educational purposes only and is not intended as medical advice, diagnosis, or treatment. Always consult your healthcare provider before making changes to your diet, supplements, or treatment plan.
References
Dong H, Zhang X, Qian Y. Mast cells and neuroinflammation. Med Sci Monit Basic Res. 2014;20:200-206. PMID: 25436584.
Zhang X, Wang Y, Dong H, Xu Y, Zhang S. Induction of microglial activation by mediators released from mast cells. Cell Physiol Biochem. 2016;38(4):1520-1531. PMID: 27050634.
Galea I. The blood-brain barrier in systemic infection and inflammation. Cell Mol Immunol. 2021;18(11):2489-2501. PMID: 34497379.
Savitz J. The kynurenine pathway: a finger in every pie. Mol Psychiatry. 2020;25(1):131-147. PMID: 30980044.
Krysiak R, Kowalcze K, Okopien B. Nickel allergy as a cause of growth hormone deficiency. Endocrine. 2021;72(3):868-874. PMID: 33419306.
Braga M, Quecchia C, Perotta C, et al. Systemic nickel allergy syndrome: nosologic framework and usefulness of diet regimen for diagnosis. Int J Immunopathol Pharmacol. 2013;26(3):707-716. PMID: 24067467.
Shimazu T, Hirschey MD, Newman J, et al. Suppression of oxidative stress by beta-hydroxybutyrate, an endogenous histone deacetylase inhibitor. Science. 2013;339(6116):211-214. PMID: 23223453.
Fortier M, Castellano CA, Croteau E, et al. A ketogenic drink improves cognition in mild cognitive impairment: results of a 6-month RCT. Alzheimers Dement. 2021;17(3):543-552. PMID: 33103819.
Breit S, Kupferberg A, Rogler G, Hasler G. Vagus nerve as modulator of the brain-gut axis in psychiatric and inflammatory disorders. Front Psychiatry. 2018;9:44. PMID: 29593576.
Ricciardi L, Minciullo PL, Saitta S, et al. Oral nickel challenge and quality of life in patients with Systemic Nickel Allergy Syndrome. Int J Immunopathol Pharmacol. 2013;26(2):449-458. PMID: 23755760.