Mast Cell Activation Syndrome and Nickel Allergy: The Inflammatory Intersection
If you have both MCAS and a nickel allergy — or if you suspect the two are connected — you're not imagining things. The biological overlap between these conditions is real, documented, and clinically important.
And if you've ever wondered why you react to high-nickel foods within *minutes* when nickel is supposed to be a "delayed" allergy — this post explains exactly why.
Two Conditions, One Inflamed Patient
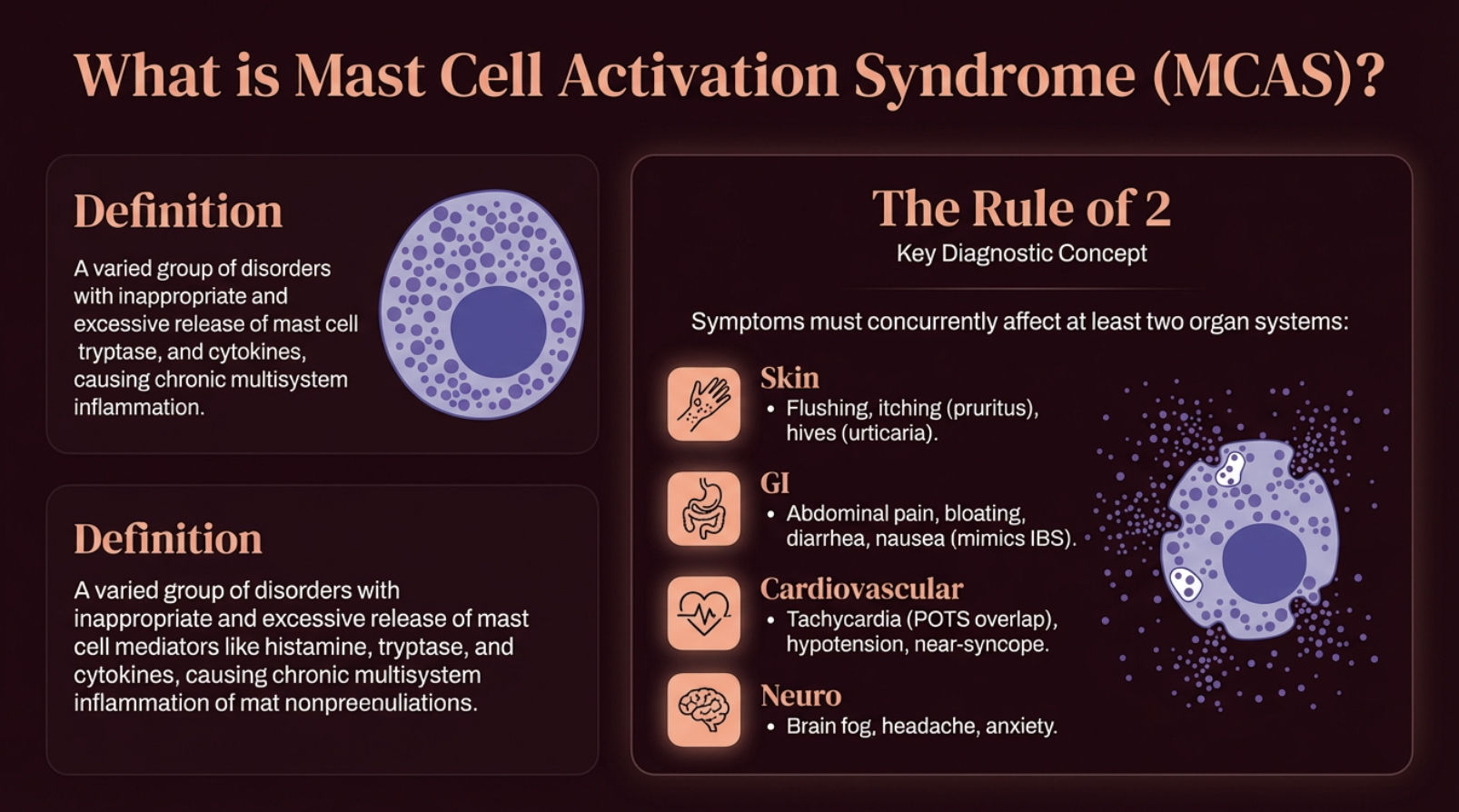
**Mast Cell Activation Syndrome (MCAS)** is characterized by the inappropriate and excessive release of mast cell mediators — histamine, tryptase, leukotrienes, and cytokines — causing chronic inflammation across multiple organ systems. To meet diagnostic criteria, symptoms must affect at least two organ systems concurrently: skin (flushing, itching, hives), GI (bloating, pain, diarrhea), cardiovascular (tachycardia, POTS overlap), and neurological (brain fog, headache, anxiety) (Weiler et al., 2019; Valent et al., 2019).
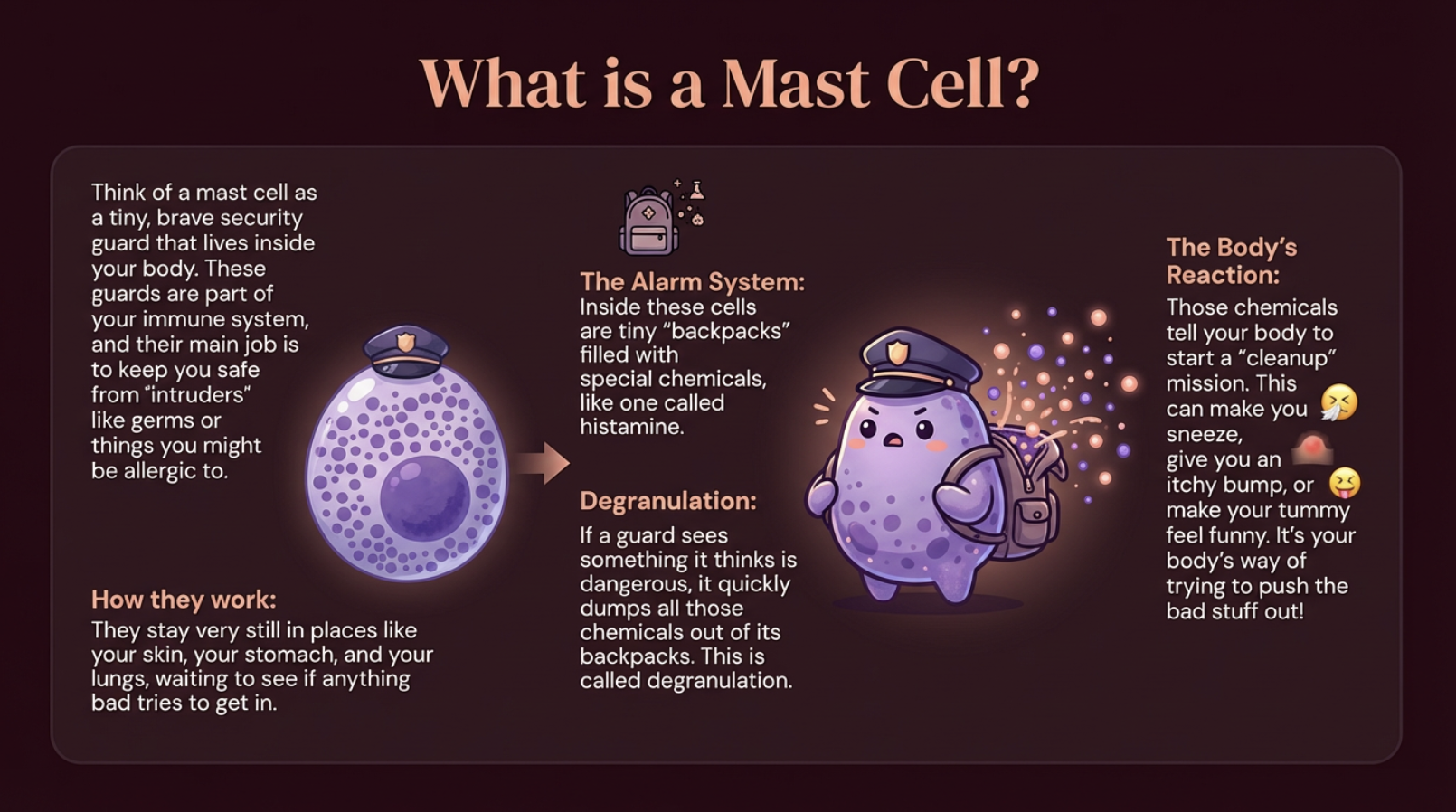
What is a mast cell?
What is mast cell activation syndrome? (MCAS)
Systemic Nickel Allergy Syndrome (SNAS) is a systemic immune response to ingested nickel. The clinical picture overlaps heavily with MCAS — dyshidrotic eczema, IBS-like GI symptoms, fatigue, headache, and brain fog (Rizzi et al., 2017; Stanghellini et al., 2016).
Read that symptom list again. The overlap is striking — and it's not coincidental.
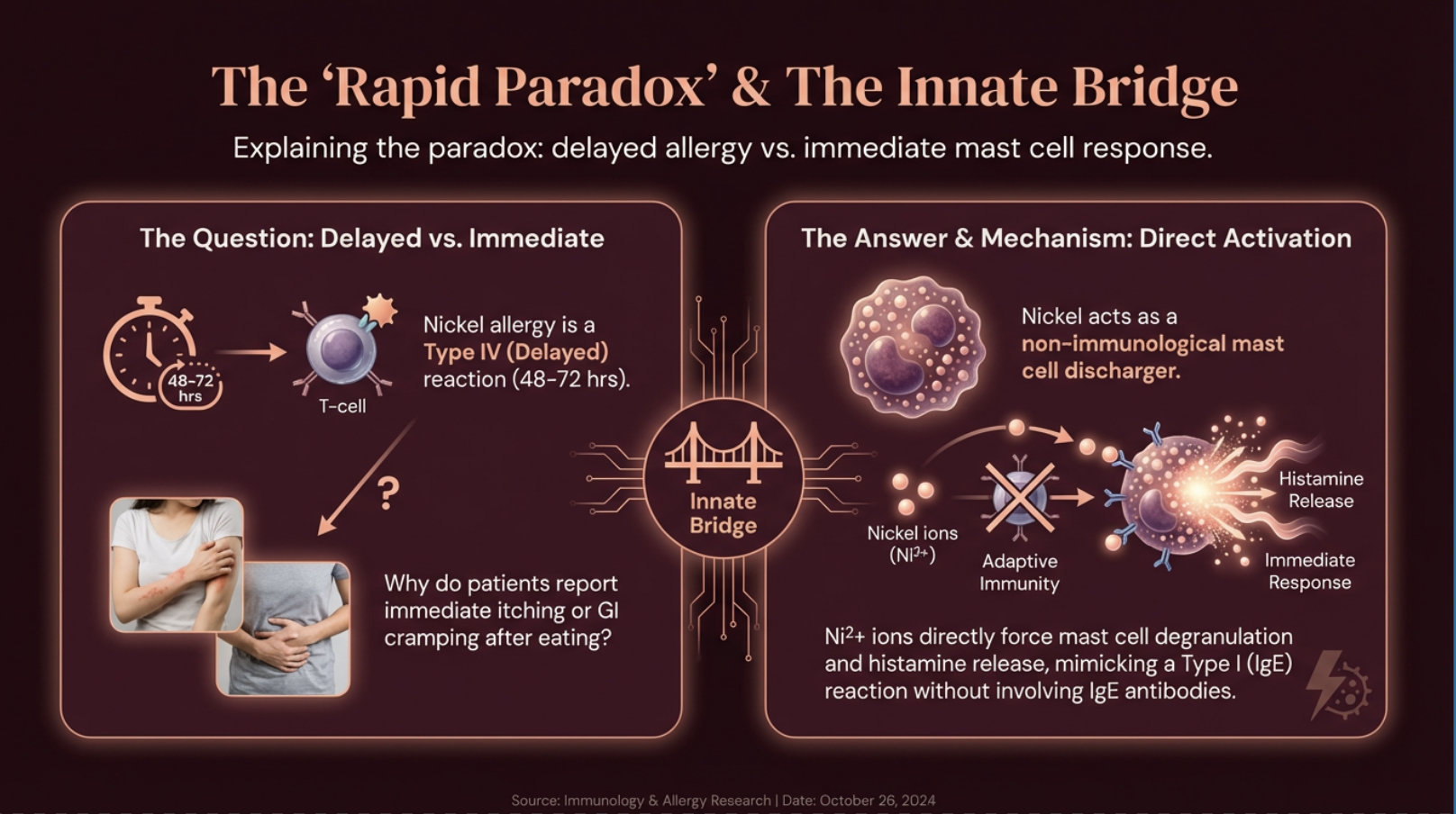
The Rapid Paradox: Why You React in Minutes
How Nickel causes immediate mast cell response alone, without a mediator like IgE
Nickel allergy is classified as a Type IV delayed hypersensitivity — meaning reactions should take 48 to 72 hours. So why do patients report immediate itching, flushing, or GI cramping after eating high-nickel foods?
Because nickel is also a **non-immunological mast cell discharger** (Walsh et al., 2010). Nickel ions can bypass the adaptive immune system entirely and directly force mast cells to degranulate, releasing histamine without requiring prior IgE-mediated sensitization (Kinbara et al., 2016).
This means nickel triggers *both* pathways simultaneously — an immediate histamine dump (minutes) AND a delayed T-cell-mediated reaction (days). You're getting hit twice from the same food.
TLR4: The Shared Trigger
Here's where the mechanisms converge. Nickel is unique among metals because it binds directly to **Toll-like Receptor 4 (TLR4)** on mast cells and endothelial cells — specifically at histidine residues H456 and H458 (Schmidt et al., 2010).
This triggers the NF-κB inflammatory pathway, resulting in rapid release of pro-inflammatory cytokines (IL-1β, IL-6) and "alarmins" (IL-33) that put mast cells into a state of hyper-reactivity — a process called **priming**.
The clinical implication is profound: chronic nickel exposure keeps your mast cells in a primed state, lowering the threshold for MCAS reactions to *all* triggers — not just nickel. Stress, heat, smells, and other foods all become more reactive because nickel has already loaded the gun.
The Angiogenesis Loop: How Inflammation Travels
Inflammation needs a highway to travel from the gut to the skin. Nickel-activated mast cells provide one by releasing **VEGF** (Vascular Endothelial Growth Factor), which builds new capillaries and lymphatic vessels (Palenca et al., 2026).
These new vessels transport immune cells from the intestinal lining to the skin, which is one mechanism behind the systemic dermatitis that SNAS patients experience. It's not just inflammation — it's inflammation that builds its own infrastructure.
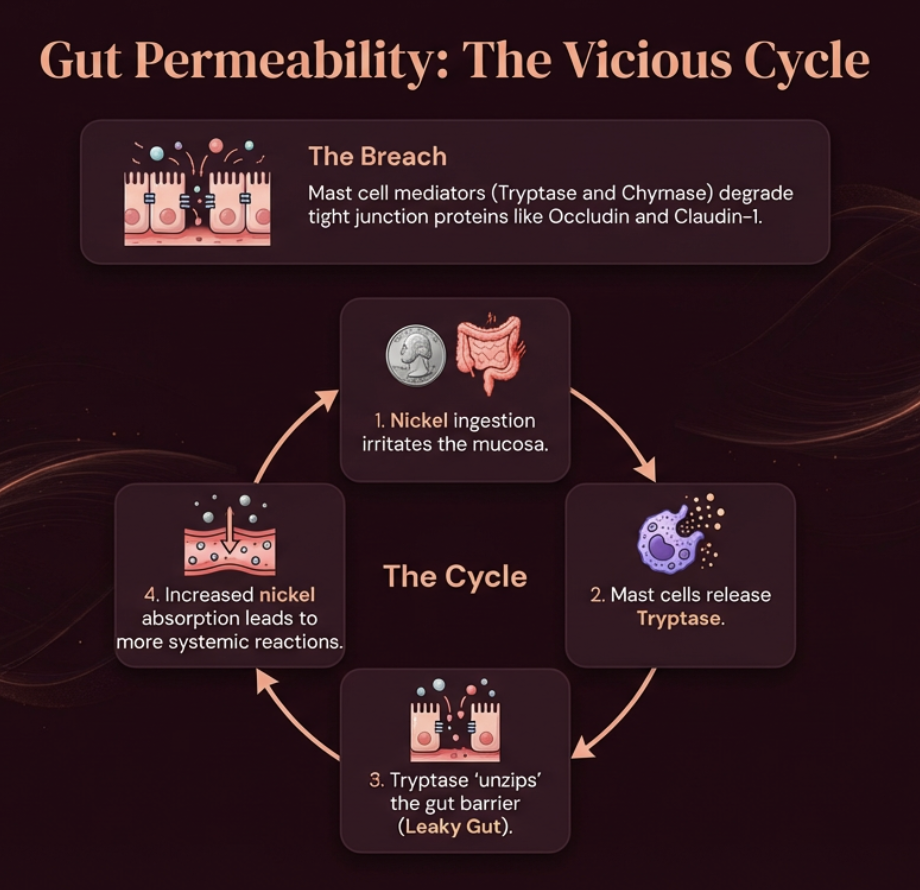
The Leaky Gut Vicious Cycle
Why gut permeability and nickel increase mast cell tryptase and histamine release
Mast cell mediators — particularly tryptase and chymase — physically degrade tight junction proteins (occludin and claudin-1) in the gut lining (Miglietta et al., 2021).
This creates a vicious cycle: nickel ingestion irritates the mucosa → mast cells release tryptase → tryptase "unzips" the gut barrier → more nickel enters the bloodstream → more systemic reactions → more mast cell activation. Each cycle makes the next one worse.
This is why addressing gut permeability is foundational for patients with both conditions — and why a low nickel diet alone, without gut repair, sometimes isn't enough.
Connected, But Distinct
Having SNAS does not guarantee MCAS, and vice versa. The distinguishing features matter:
**SNAS** symptoms are dose-dependent on nickel intake. Reactions are often delayed (except for the innate mast cell trigger). Confirmed by patch test or dietary elimination.
**MCAS** symptoms are triggered by many factors — stress, heat, vibration, smells, and a wide range of foods. Reactions are often immediate or anaphylactoid. Diagnosed by lab evidence (elevated tryptase, urinary histamine metabolites) plus response to mast cell-targeted therapy.
**The overlap:** the "primed patient" — someone with high baseline inflammation and gut barrier compromise where nickel is one of multiple triggers keeping the immune system in a state of constant activation.
The critical clinical insight: untreated nickel allergy can *worsen* MCAS by keeping the immune system chronically inflamed, lowering the threshold for reactions to everything else. Removing dietary nickel may not cure MCAS — but it removes a significant inflammatory load that makes every other trigger worse.
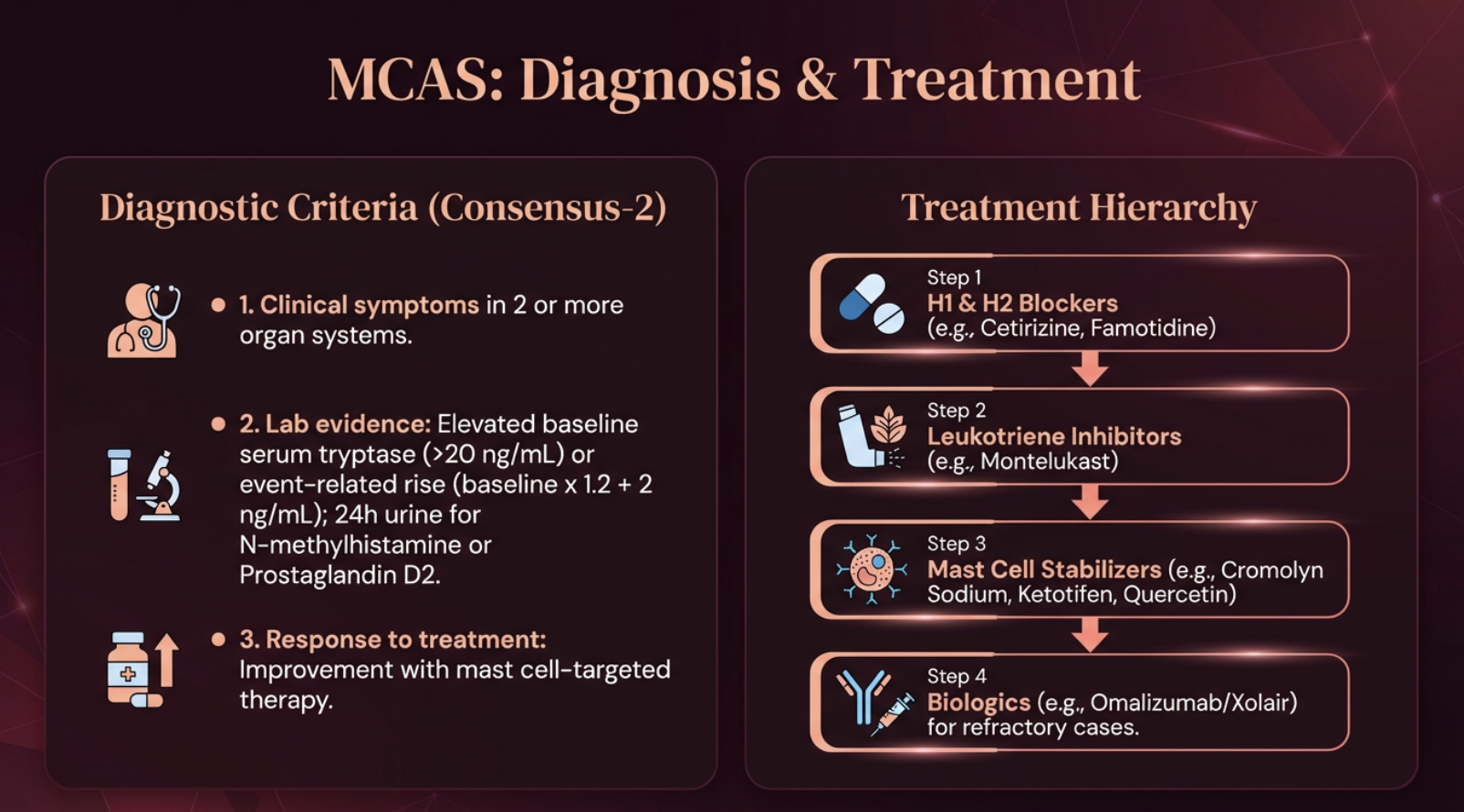
MCAS Diagnosis: The Current Framework
The Consensus-2 diagnostic criteria require three elements (Weiler et al., 2019; Valent et al., 2019):
**1. Clinical symptoms** in two or more organ systems.
**2. Lab evidence:** Elevated baseline serum tryptase (>20 ng/mL) or an event-related rise (baseline × 1.2 + 2 ng/mL). Also 24-hour urine for N-methylhistamine or Prostaglandin D2.
**3. Response to treatment:** Improvement with mast cell-targeted therapy.
The treatment hierarchy follows a stepwise approach: H1 and H2 blockers (cetirizine, famotidine) → leukotriene inhibitors (montelukast) → mast cell stabilizers (cromolyn sodium, ketotifen, quercetin) → biologics (omalizumab/Xolair) for refractory cases.
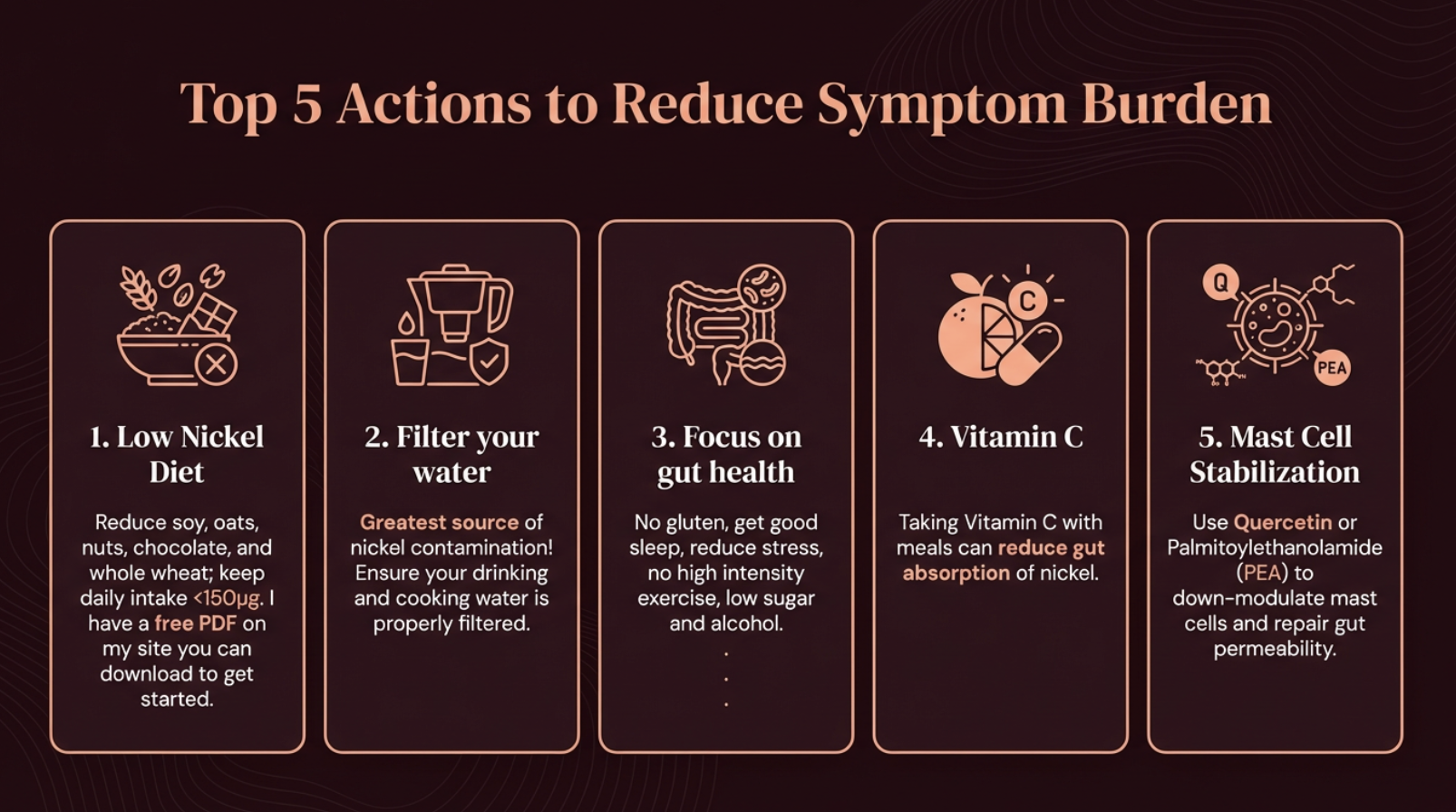
5 Actions to Reduce Your Symptom Burden
Top 5 actions to reduce MCAS symptom burden
**1. Low nickel diet.** Reduce the highest offenders — soy, oats, nuts, chocolate, whole wheat. Keep daily intake below 150 mcg. This removes one of the most potent mast cell-priming triggers from your system.
**2. Flush the pipes.** Run tap water for 30–60 seconds in the morning. Nickel leaches from faucet fixtures overnight, and absorption from water on an empty stomach is 40 times higher than from food (Solomons et al., 1982).
**3. Cookware audit.** Switch from stainless steel to glass, ceramic, or cast iron — especially for acidic foods like tomato sauce, which accelerate nickel leaching.
**4. Mast cell stabilization.** Consider quercetin or Palmitoylethanolamide (PEA). PEA acts as a "gatekeeper," down-modulating mast cells and supporting gut barrier repair (Palenca et al., 2026).
**5. Vitamin C with meals.** Binds to nickel in the gut, reducing how much enters your bloodstream and reducing the mast cell trigger at its source (Solomons et al., 1982).
Your Next Step
If you have MCAS and haven't been evaluated for nickel allergy — or if you have nickel allergy and your symptoms seem disproportionate to your dietary nickel intake — the overlap is worth exploring. Start with the diet and see what shifts.
Download my **free Nickel Food List** to identify the highest-nickel foods in your current diet.
**[Download the Free Nickel Food List →](https://the-low-nickel-diet.kit.com/foodlists)**
---
References
Afrin LB, Self S, Menk J, Lazarchick J. Characterization of Mast Cell Activation Syndrome. Am J Med Sci. 2017;353(3):207-215.
Antico A, Soana R. Nickel sensitization and dietary nickel are a substantial cause of symptoms provocation in patients with chronic allergic-like dermatitis syndromes. Allergy Rhinol (Providence). 2015;6(1):e56-e63.
Bergman D, Goldenberg A, Rundle C, Jacob SE. Low Nickel Diet: A Patient-Centered Review. J Clin Exp Dermatol Res. 2016;7(3):355.
Borghini R, Porpora MG, Casale R, et al. Irritable Bowel Syndrome-Like Disorders in Endometriosis: Prevalence of Nickel Sensitivity and Effects of a Low-Nickel Diet. An Open-Label Pilot Study. Nutrients. 2020;12(2):341.
Di Tola M, Marino M, Amodeo R, et al. Immunological characterization of the allergic contact mucositis related to the ingestion of nickel-rich foods. Immunobiology. 2014;219(7):522-530.
Kinbara M, Bando K, Shiraishi D, et al. Mast cell histamine-mediated transient inflammation following exposure to nickel promotes nickel allergy in mice. Exp Dermatol. 2016;25(6):466-471.
Miglietta S, Borghini R, Relucenti M, et al. New Insights into Intestinal Permeability in Irritable Bowel Syndrome-Like Disorders: Histological and Ultrastructural Findings of Duodenal Biopsies. Cells. 2021;10(10):2593.
Molderings GJ, Haenisch B, Brettner S, et al. Pharmacological treatment options for mast cell activation disease. Naunyn Schmiedebergs Arch Pharmacol. 2016;389(7):671-694.
Palenca I, Basili Franzin S, Sarnelli G, Esposito G. Palmitoylethanolamide for Nickel Allergy: Plausible, Untested, and Worth Considering. Biomedicines. 2026;14(1):177.
Picarelli A, Di Tola M, Vallecoccia A, et al. Oral Mucosa Patch Test: A New Tool to Recognize and Study the Adverse Effects of Dietary Nickel Exposure. Biol Trace Elem Res. 2011;139(2):151-159.
Rizzi A, Nucera E, Laterza L, et al. Irritable Bowel Syndrome and Nickel Allergy: What Is the Role of the Low Nickel Diet? J Neurogastroenterol Motil. 2017;23(1):101-108.
Schmidt M, Raghavan B, Müller V, et al. Crucial role for human Toll-like receptor 4 in the development of contact allergy to nickel. Nat Immunol. 2010;11(9):814-819.
Sharma AD. Low nickel diet in dermatology. Indian J Dermatol. 2013;58(3):240-247.
Solomons NW, Viteri F, Shuler TR, Nielsen FH. Bioavailability of nickel in man: effects of foods and chemically-defined dietary constituents on the absorption of inorganic nickel. J Nutr. 1982;112(1):39-50.
Stanghellini V, Tosetti C, Benedetto E, et al. Nickel sensitization in patients with gastro-esophageal reflux disease. United European Gastroenterol J. 2016;4(2):184-190.
Valent P, Akin C, Bonadonna P, et al. Proposed Diagnostic Algorithm for Patients with Suspected Mast Cell Activation Syndrome. J Allergy Clin Immunol Pract. 2019;7(4):1125-1133.
Walsh ML, Smith VH, King CM. Type 1 and type IV hypersensitivity to nickel. Australas J Dermatol. 2010;51(4):285-286.
Weiler CR, Austen KF, Akin C, et al. AAAAI Mast Cell Disorders Committee Work Group Report: Mast cell activation syndrome (MCAS) diagnosis and management. J Allergy Clin Immunol. 2019;144(4):883-896.