Surgical & Dental Implants & Systemic Nickel Allergy: A Better Framework for the Decision
If you have nickel allergy and you need a surgical or dental implant, or you already have one and you have started to wonder whether it might be making you sick, you are working with a question that the medical literature has not fully answered. Your surgeon is working with the best information they have. That information is more incomplete than they may realize. I am Dr. Laura Duzett, a resident physician training in pathology and a Nutritional Therapy Practitioner. I have systemic nickel allergy syndrome (SNAS) myself.
After two months of assessing the implant-allergy literature, I came away convinced of three things that I think every patient and surgeon facing this decision should know.
The first is that the science of how nickel meets the immune system has matured enormously in the last fifteen years.
The second is that the testing protocols used in the published implant studies have not caught up with the science.
The third is that we now know enough to do this better.
This article walks through the immunology, what each test can and cannot prove, what the watershed 2025 randomized trial actually showed, and a more targeted workup framework you can bring to your care team. The free companion guide, linked at the bottom, includes a doctor's letter you can hand to your surgeon and the full clinical reference set.
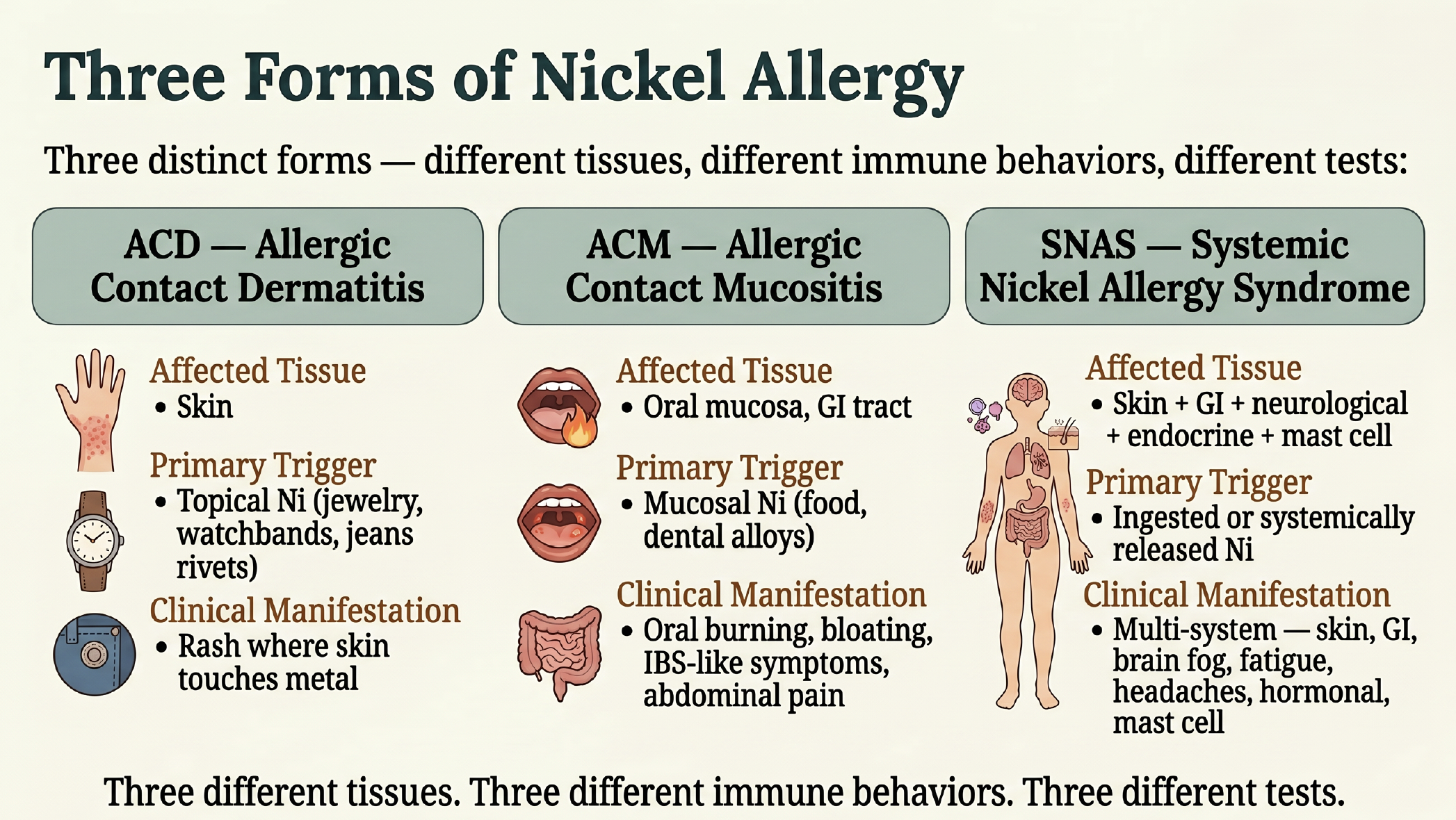
The three forms of nickel allergy
Three distinct forms of allergy with different immune cells and behaviors, requiring DIFFERENT TESTS!
Nickel allergy is not one thing. It is three different things, in three different tissues, with three different immune behaviors, and three different tests.
**Allergic contact dermatitis (ACD)** is the classic skin reaction to topical nickel. Earrings, watch backs, jeans rivets. Skin contacts metal, the rash appears at the contact site within a couple of days.
**Allergic contact mucositis (ACM)** is the gut version. Instead of a rash on the skin, the lining of the mouth and the gut reacts to ingested nickel. Symptoms look like bloating, abdominal pain, oral burning, and what is often diagnosed as IBS. If you want the deeper dive on that overlap, the IBS and Systemic Nickel Allergy article walks through it.
**Systemic nickel allergy syndrome (SNAS)** is the whole-body version. This is when nickel reaches the bloodstream and triggers reactions across multiple tissue compartments simultaneously: skin, gut, brain (fog, fatigue, headaches), endocrine, and mast cell. Most people watching my live streams are dealing with some degree of SNAS even if they have never been formally diagnosed.
The What is SNAS primer is the place to start if any of this is new. Three different tissues. Three different immune behaviors. Three different tests. That is the foundation for everything else.
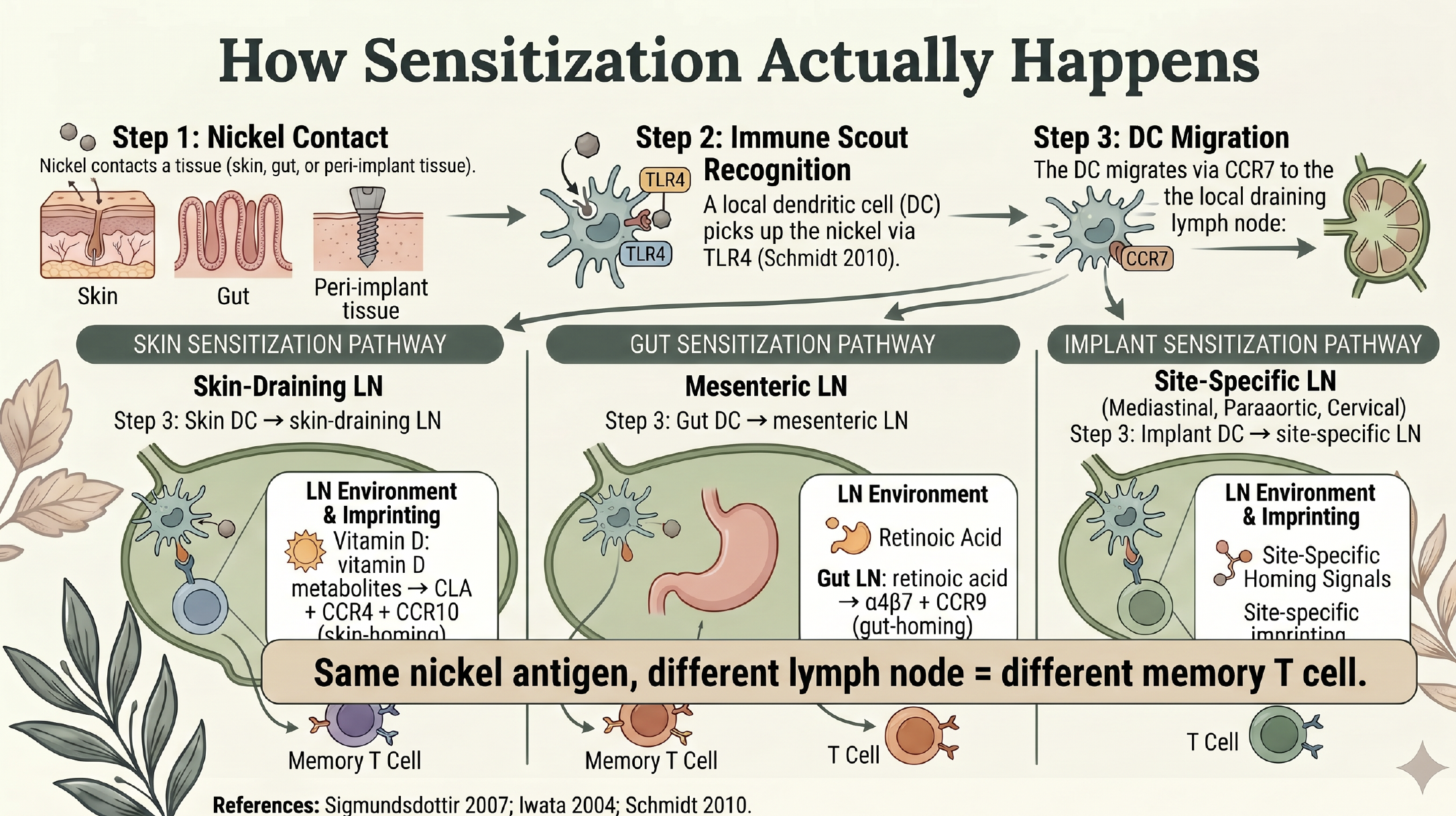
How sensitization actually happens
The steps of sensitization in the three different types
When nickel ions contact a tissue, a local dendritic cell — the immune system's reconnaissance scout — picks them up using a pattern-recognition receptor called Toll-like receptor 4 (Schmidt 2010, PMID 20835230). The dendritic cell then migrates via the chemokine receptor CCR7 to the draining lymph node nearest to where it was sampling. Here is the part that matters. Inside the lymph node, the dendritic cell presents the nickel to a naive T cell, and the local lymph node environment imprints that T cell with homing receptors specific to where the dendritic cell came from. Skin-draining lymph nodes use vitamin D metabolites to imprint CLA, CCR4, and CCR10 — the skin-homing receptors. Gut-draining lymph nodes use retinoic acid to imprint α4β7 integrin and CCR9 — the gut-homing receptors. Same nickel antigen. Different lymph node. Completely different memory T cell. After activation, these memory T cells split into three pools.
**Tissue-resident memory cells (TRM)** stay locked in the originating tissue and never circulate — they are the local sentinels.
**Effector memory cells (TEM)** travel through non-lymphoid tissues but only patrol the type of tissue their badge specifies.
**Central memory cells (TCM)** circulate through the lymph node system as the long-term coordinators.
This is the cell-level reason a single implant can produce skin, gut, and brain symptoms simultaneously. The implant feeds nickel into the bloodstream through low-grade ion release. Pre-existing memory T cells from past sensitization (most patients with nickel allergy already have skin-imprinted memory from old jewelry exposure) get reactivated. New peri-implant dendritic cells migrate to local lymph nodes and create new memory pools. Biliary excretion sends nickel back into the gut lumen, where it can drive mucosal mast cell activation and tight-junction degradation. Sweat excretion concentrates nickel at the palms and soles. The mechanism behind that last route is the Dyshidrotic Eczema and Systemic Nickel Allergy story in detail. One source. Multiple tissues activated.
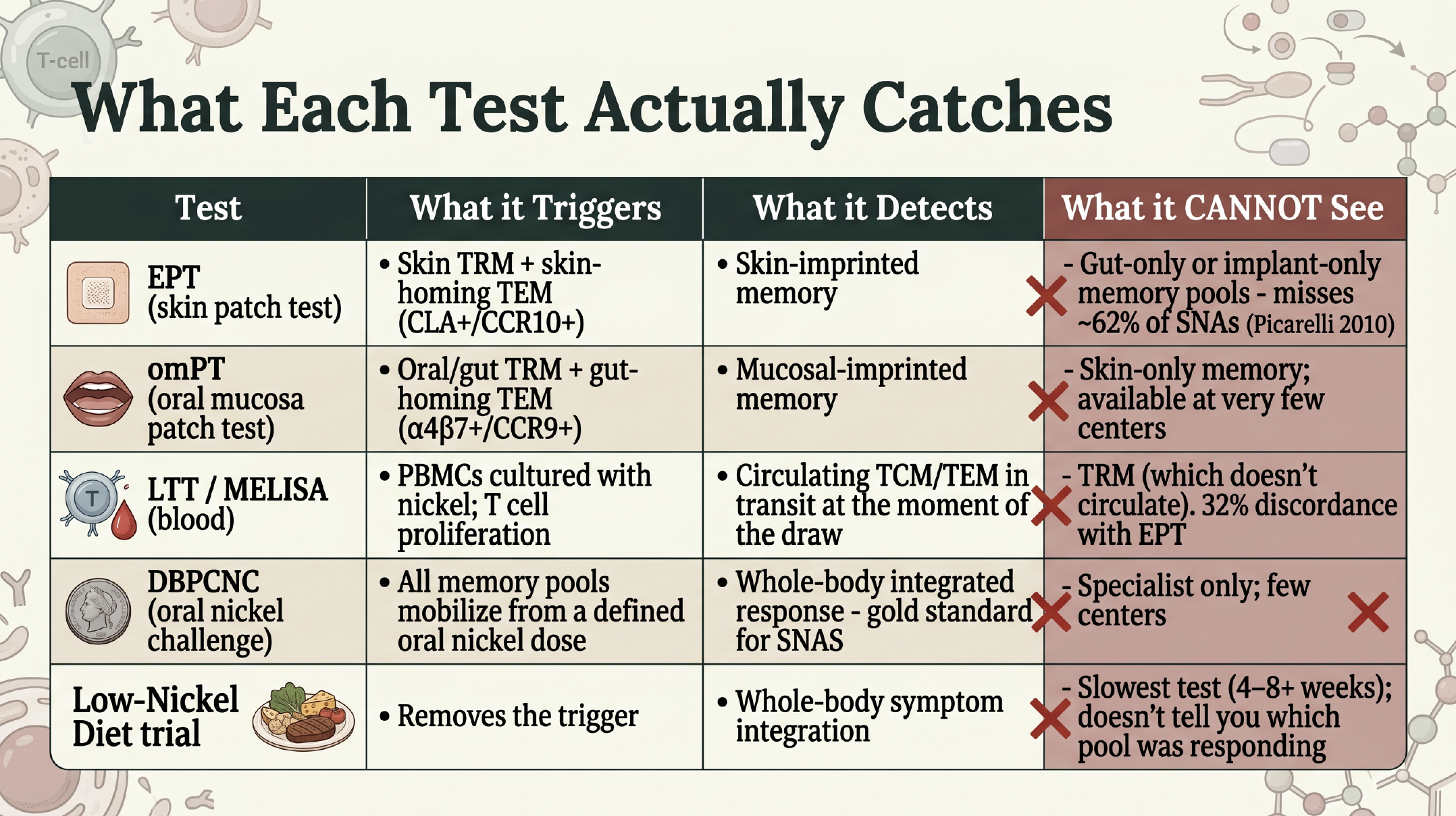
What each test actually catches — and what it cannot
This is where the immunology starts to do practical work. Each test in the available toolkit catches a different cell population.
The limitations of each test for nickel allergy
The deeper version of this discussion lives in the SNAS Testing article, and the conversation I had with Dr. Matthew Zirwas — one of the few American dermatologists who runs this work clinically — is in the Dr. Zirwas interview. The headline takeaway is simple. A negative skin patch test does not rule out SNAS. Picarelli's 2010 paper put numbers on it directly: in 86 symptomatic patients, the skin patch test was positive in 38.4%; the oral mucosa patch test was positive in 61.6% (P=0.004). Different tissue, nearly double the detection rate.
The watershed paper: Apostolos 2025 INSPIRE
In April 2025, *Circulation: Cardiovascular Interventions* published the first prospective randomized trial in the entire implant-allergy literature (Apostolos et al., PMID 40057986). Ninety-six patients undergoing PFO closure were randomized to either Amplatzer or Gore Cardioform devices and followed for ninety days. They were stratified by pre-procedure skin patch test result. The nickel-allergic subgroup had a device syndrome rate of 71.4% versus 20.6% in the non-allergic group. Adjusted odds ratio: 10.53 (95% CI 3.17 to 35.00, P<0.001). New or worsening migraine: 21.4% versus 1.5%. Palpitations: 50.0% versus 14.7%. The trial had no industry funding — funded entirely by the non-profit Hellenic Society of Cardiology. This is mainstream cardiology formally measuring what cardiologists historically dismissed. There is, however, a structural caveat the trial itself does not address. The Apostolos trial used the skin patch test to define who was nickel-allergic. Given what the immunology shows about skin versus gut versus circulating memory pools, the trial is almost certainly under-counting allergy in its non-allergic group, and the real signal is probably even larger than what the trial captured.
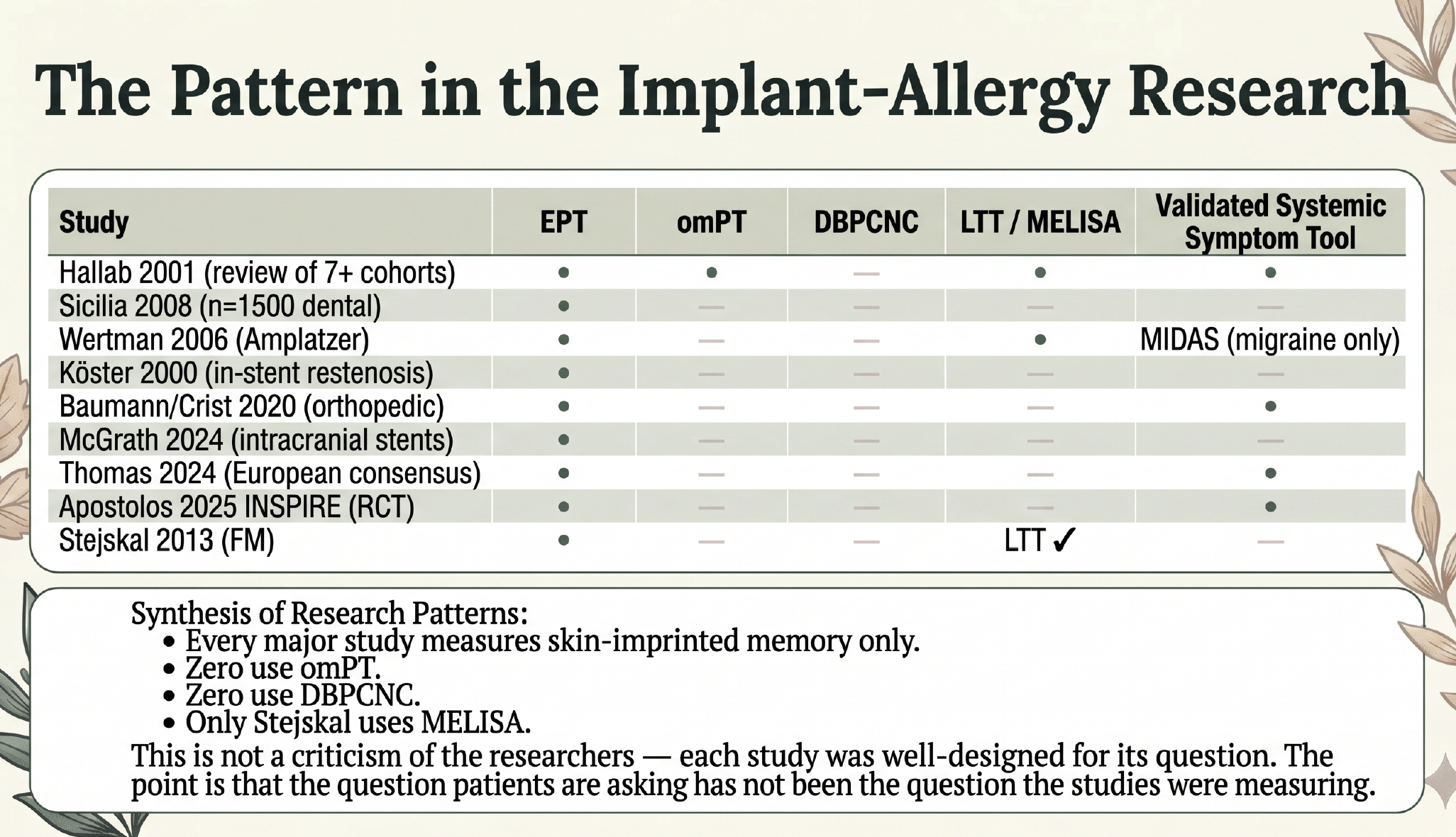
The methodologic monoculture
Once you tabulate the implant-allergy literature against the immunology, a striking pattern appears.
The tests used to characterize whether or not nickel hypersensitivity risk or reality was a factor were not the right tests to answer those questions in almost all of these papers!
Every major implant-allergy study has measured skin-imprinted memory only. Zero studies use the oral mucosa patch test. Zero use the oral nickel challenge. None use validated systemic symptom instruments — no IBS-SSS, no FACIT-Fatigue, no validated MCAS criteria. This is not a criticism of the researchers.
The point is that the field has been answering one question (skin reactivity) and inferring something different (systemic disease) from the answer. The mechanism does not support that inference.
A useful way to think about this: imagine a state police force that only ever runs sting operations in the skin neighborhood. If your gang of bad actors is operating mostly in the gut, or running an implant compound that distributes through the bloodstream and the bile and the sweat, the skin sting comes back negative — and you get told there is no gang problem in your city. The activity is real. It is just not where the surveillance is set up.
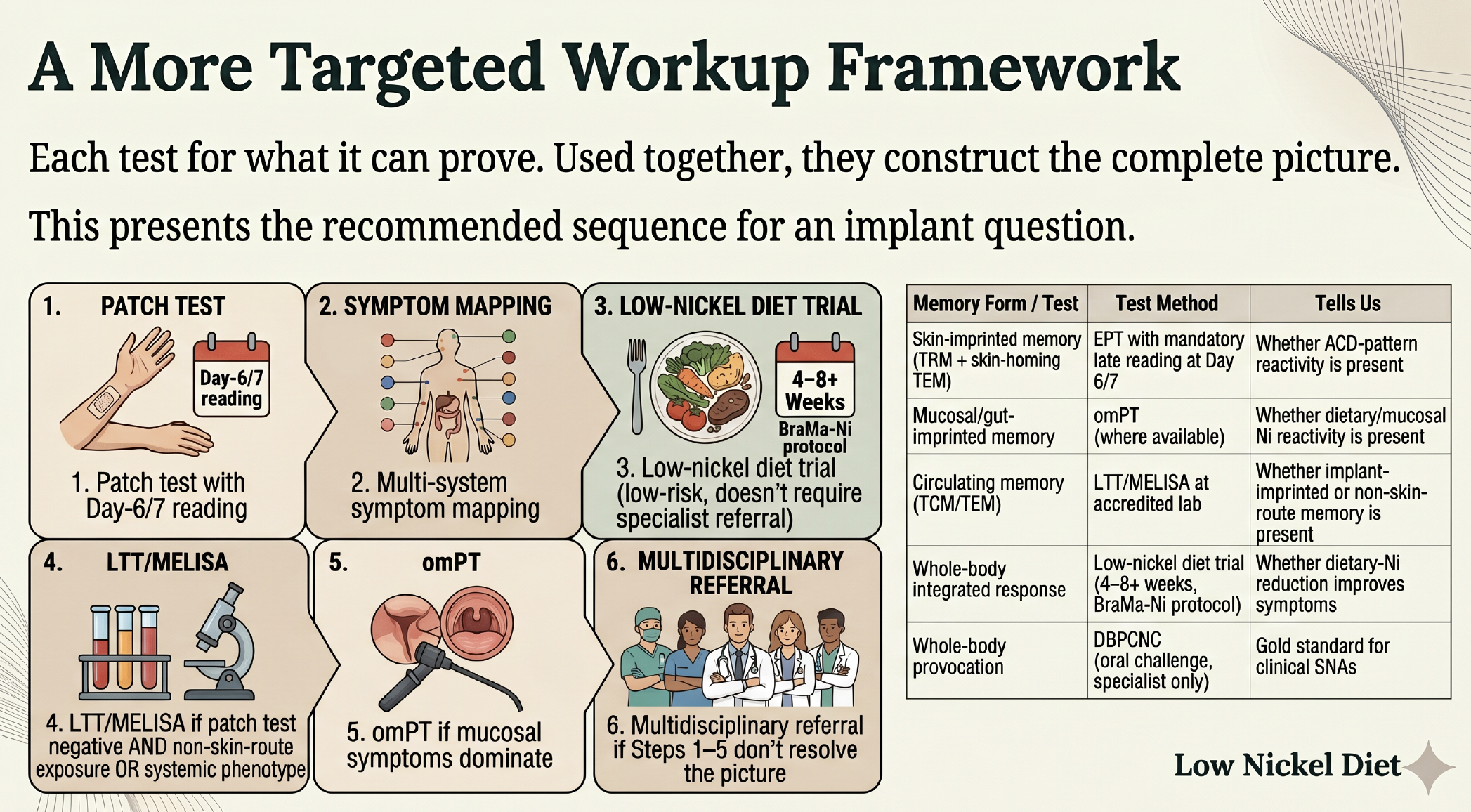
A more targeted workup framework
The constructive answer is that each test should be used for what it can prove, and the pieces combined to give a real picture. For someone facing an implant decision, or already implanted with new systemic symptoms, the proposed sequence is:
1. **Patch test with mandatory Day 6/7 reading.** First line, accessible. Tells you whether ACD-pattern reactivity is present.
2. **Multi-system symptom mapping.** Skin, gut, neurological, fatigue, mast cell. Not just the local site.
3. **Low-nickel diet trial.** Four to eight weeks using the BraMa-Ni protocol. Low-risk, mechanistically grounded, and does not require a specialist referral. The What is the Low Nickel Diet primer covers the basics.
4. **LTT/MELISA at an accredited lab** if the patch test is negative AND the implant exposure is non-skin-route, or if the symptoms are systemic.
5. **omPT at a specialty center** if mucosal symptoms dominate.
6. **Multidisciplinary referral** if Steps 1 through 5 do not resolve the picture: allergy, the relevant surgical specialty, gastroenterology, and a nutrition-aware physician or NTP.
Source reduction has evidence. Chelation does not. The Stejskal 2013 fibromyalgia data — the closest thing in the literature to a long-term metal-removal outcome study — was a source-reduction protocol (dental amalgam replacement, not chelation). The fibromyalgia overlap with metal allergy is a much larger story that I covered in the Fibromyalgia and Nickel Allergy deep dive. The mast cell layer is in MCAS and Systemic Nickel Allergy.
How to talk to your surgeon
Bring patch test results with reagent concentrations and grading. Bring your LTT/MELISA result if you have one. Bring an allergist letter if you have one. Bring the doctor's letter from the free guide. Ask what alloy the surgeon typically uses. Ask the percent nickel content. Ask whether nickel-free alternatives appropriate to your specific surgery exist, and what the surgeon's volume is with those alternatives. Ask what the long-term revision rate looks like (alternatives are not always free — Australian Joint Registry data shows roughly two times higher 12-year revision risk for Oxinium knees, for example). Ask what documentation you need for insurance pre-authorization. Frame the conversation collaboratively. Your goal is the same as theirs: the best decision for your specific situation. Bringing updated literature to a surgical consult is part of being a partner in your own care.
The guide
If you would like the doctor's letter, the printable testing checklists, the device-specific reference table with manufacturer details, and the full citation list with PMIDs, the free companion guide — *The Implant & Nickel Allergy Decision Guide* — is available in the private community.
The doctor's letter is written in collaborative, professional language. It is designed to make the conversation easier, not adversarial. Download the free guide in the private community.
Related reading
- SNAS Testing — What Actually Works
- Dyshidrotic Eczema and Systemic Nickel Allergy
- Dr. Matthew Zirwas Interview — Nickel Allergy From Food
- MCAS and Systemic Nickel Allergy
- Fibromyalgia and Nickel Allergy
References
1. Apostolos A, Drakopoulou M, Gregoriou S, et al. Patent foramen ovale closure in patients with and without nickel hypersensitivity: a randomized trial. *Circ Cardiovasc Interv*. 2025;18(4):e015228. PMID 40057986.
2. Picarelli A, Di Tola M, Vallecoccia A, et al. Oral mucosa patch test: a new tool to recognize and study the adverse effects of dietary nickel exposure. *Biol Trace Elem Res*. 2011;139(2):151-159. PMID 20204548.
3. Hallab N, Merritt K, Jacobs JJ. Metal sensitivity in patients with orthopaedic implants. *J Bone Joint Surg Am*. 2001;83(3):428-436. PMID 11263649.
4. Sicilia A, Cuesta S, Coma G, et al. Titanium allergy in dental implant patients: a clinical study on 1500 consecutive patients. *Clin Oral Implants Res*. 2008;19(8):823-835. PMID 18705814.
5. Subramanian S, Iyer S, Johnson G, Agrawal H, Fraser CD. Resolution of severe ulcerative colitis secondary to nickel allergy following explantation of Amplatzer septal occluder device. *World J Pediatr Congenit Heart Surg*. 2025;16(1):119-121. PMID 39285815.
6. Stejskal V, Öckert K, Bjørklund G. Metal-induced inflammation triggers fibromyalgia in metal-allergic patients. *Neuro Endocrinol Lett*. 2013;34(6):559-565. PMID 24378456.
7. Thomas P, Arenberger P, Bader R, et al. A literature review and expert consensus statement on diagnostics in suspected metal implant allergy. *J Eur Acad Dermatol Venereol*. 2024;38(8):1471-1477. PMID 38606660.
8. Wertman B, Azarbal B, Riedl M, Tobis J. Adverse events associated with nickel allergy in patients undergoing percutaneous atrial septal defect or patent foramen ovale closure. *J Am Coll Cardiol*. 2006;47(6):1226-1227. PMID 16545656.
9. Schmidt M, Raghavan B, Müller V, et al. Crucial role for human Toll-like receptor 4 in the development of contact allergy to nickel. *Nat Immunol*. 2010;11(9):814-819. PMID 20835230.
10. Sigmundsdottir H, Pan J, Debes GF, et al. DCs metabolize sunlight-induced vitamin D3 to "program" T cell attraction to the epidermal chemokine CCL27. *Nat Immunol*. 2007;8(3):285-293.
11. Iwata M, Hirakiyama A, Eshima Y, et al. Retinoic acid imprints gut-homing specificity on T cells. *Immunity*. 2004;21(4):527-538.
12. Köster R, Vieluf D, Kiehn M, et al. Nickel and molybdenum contact allergies in patients with coronary in-stent restenosis. *Lancet*. 2000;356(9245):1895-1897. PMID 11130387.
13. McGrath M, Clarke J, Midtlien JP, et al. Safety of intracranial venous stenting in patients with nickel allergy. *J Neurointerv Surg*. 2024. PMID 39084853.
14. Baumann CA, Crist BD. Nickel allergy to orthopaedic implants: a review and case series. *J Clin Orthop Trauma*. 2020;11(Suppl 4):S596-S603. PMID 32774035.
15. Braga M, Quecchia C, Perotta C, et al. Systemic nickel allergy syndrome: nosologic framework and diet regimen. *Int J Immunopathol Pharmacol*. 2013;26(3):707-716.
*This article is for educational purposes only and is not medical advice. Always consult your own healthcare team before making changes to your treatment plan.*