Is Nickel the ACTUAL cause of Histamine Intolerance?!
The Nickel-Histamine Connection: Why a Low-Histamine Diet Only Gets You 60% Better
If you've tried a low-histamine diet and felt better — but not all the way better — this post is for you.
You followed the lists. You cut out aged cheese, wine, sauerkraut, soy sauce, cured meats. Maybe you even gave up your favorite kombucha. And it helped. The bloating eased. The diarrhea improved. The acid reflux settled down.
But you still flush. You still get migraines. You still can't sleep. You still break out in hives at seemingly random times.
You're not doing anything wrong. The low-histamine diet is addressing only one piece of the puzzle — and it happens to be the smallest piece. Today I want to walk you through what the research actually shows about where your systemic histamine is coming from, why dietary histamine is the least of your worries, and how an overlooked condition called Systemic Nickel Allergy Syndrome may be attacking your histamine system at five different points simultaneously.
First: What Is Histamine, Really?
Histamine is not a villain. It's a normal, essential signaling molecule that your body needs for stomach acid production, immune defense, neurotransmission, and regulating your sleep/wake cycle. The problem isn't that histamine exists — it's that your body is accumulating more than it can break down.
Histamine intolerance (HIT) is estimated to affect 1–3% of the population, with roughly 80% of diagnosed cases occurring in middle-aged women [3, 4]. It's frequently misdiagnosed as IBS, chronic migraine, chronic urticaria, or generalized food allergy — partly because there is currently no single validated diagnostic test for HIT [4, 5].
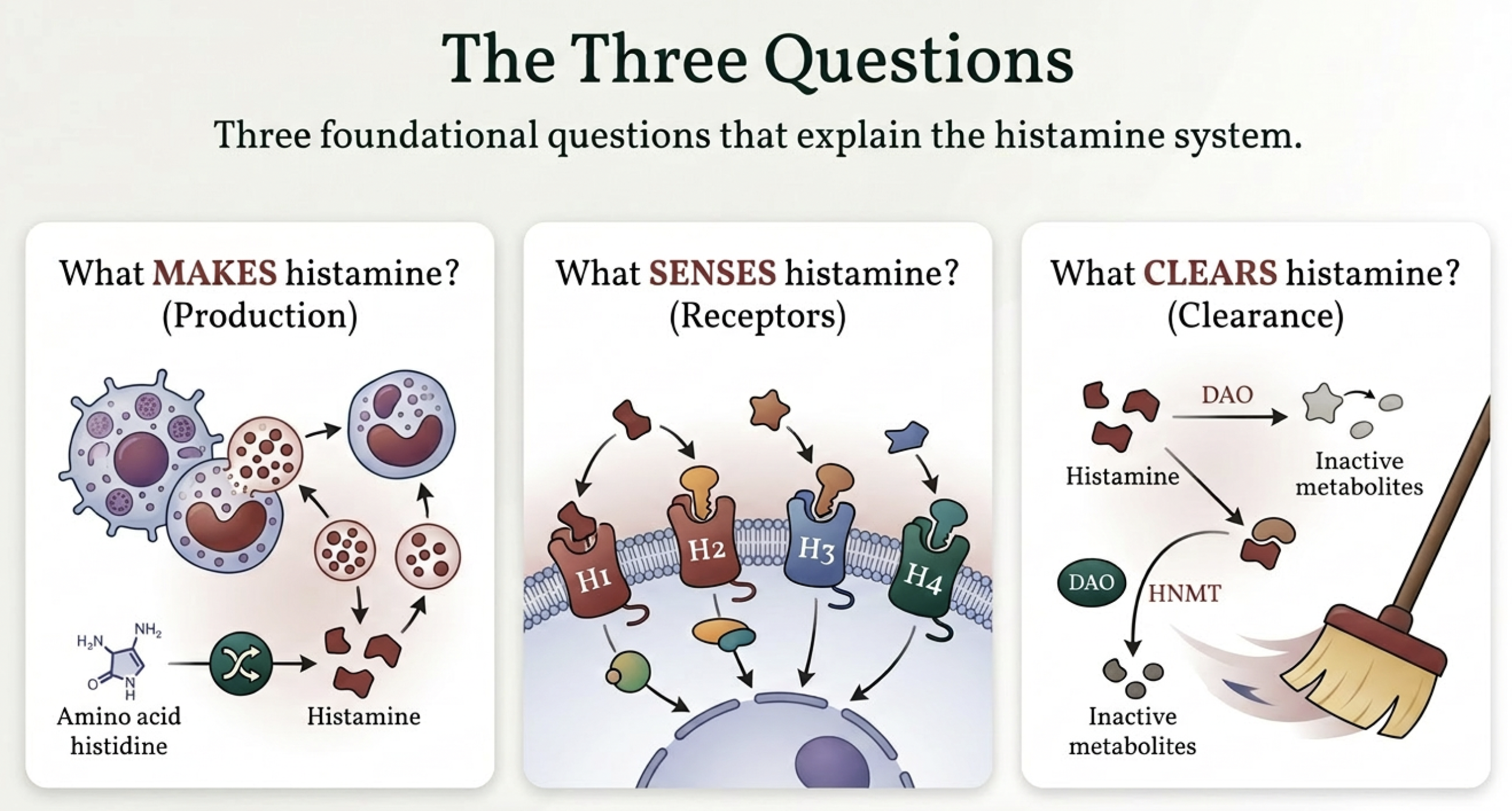
The Three Questions That Explain Everything
Every aspect of histamine intolerance comes down to three questions: What makes histamine? What senses it? And what clears it?
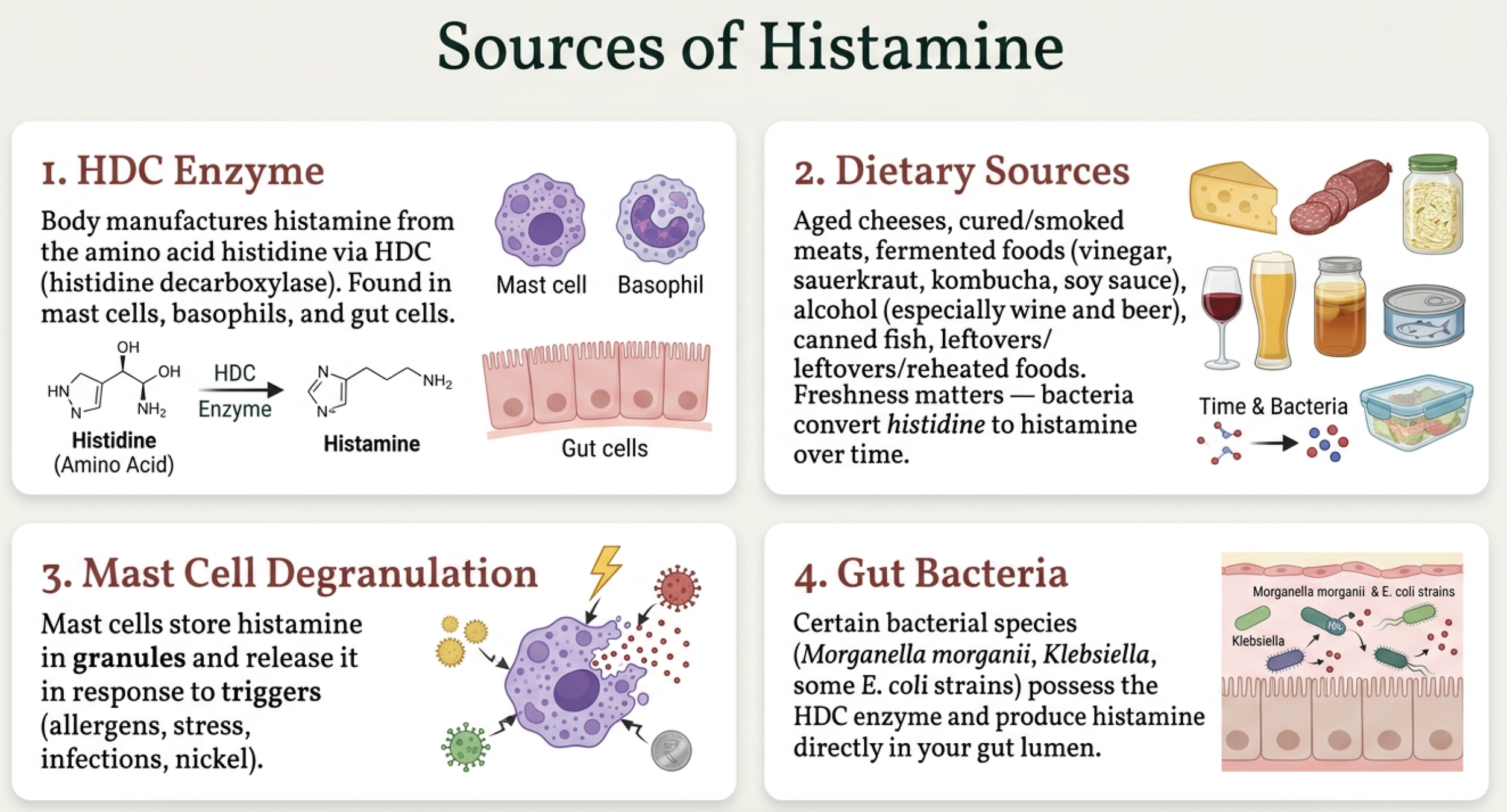
What makes histamine
Your body has four sources of histamine. Your own cells manufacture it via an enzyme called histidine decarboxylase (HDC), primarily in mast cells, basophils, and gut lining cells [3]. Your food contributes histamine when it's aged, fermented, or improperly stored — bacteria convert the amino acid histidine into histamine over time, which is why freshness matters so much. Your mast cells store histamine in granules and release it when triggered by allergens, stress, infections, or nickel [12]. And certain gut bacteria — species like Morganella morganii and Klebsiella — possess the HDC enzyme and produce histamine directly in your gut lumen [3, 5].
What senses histamine
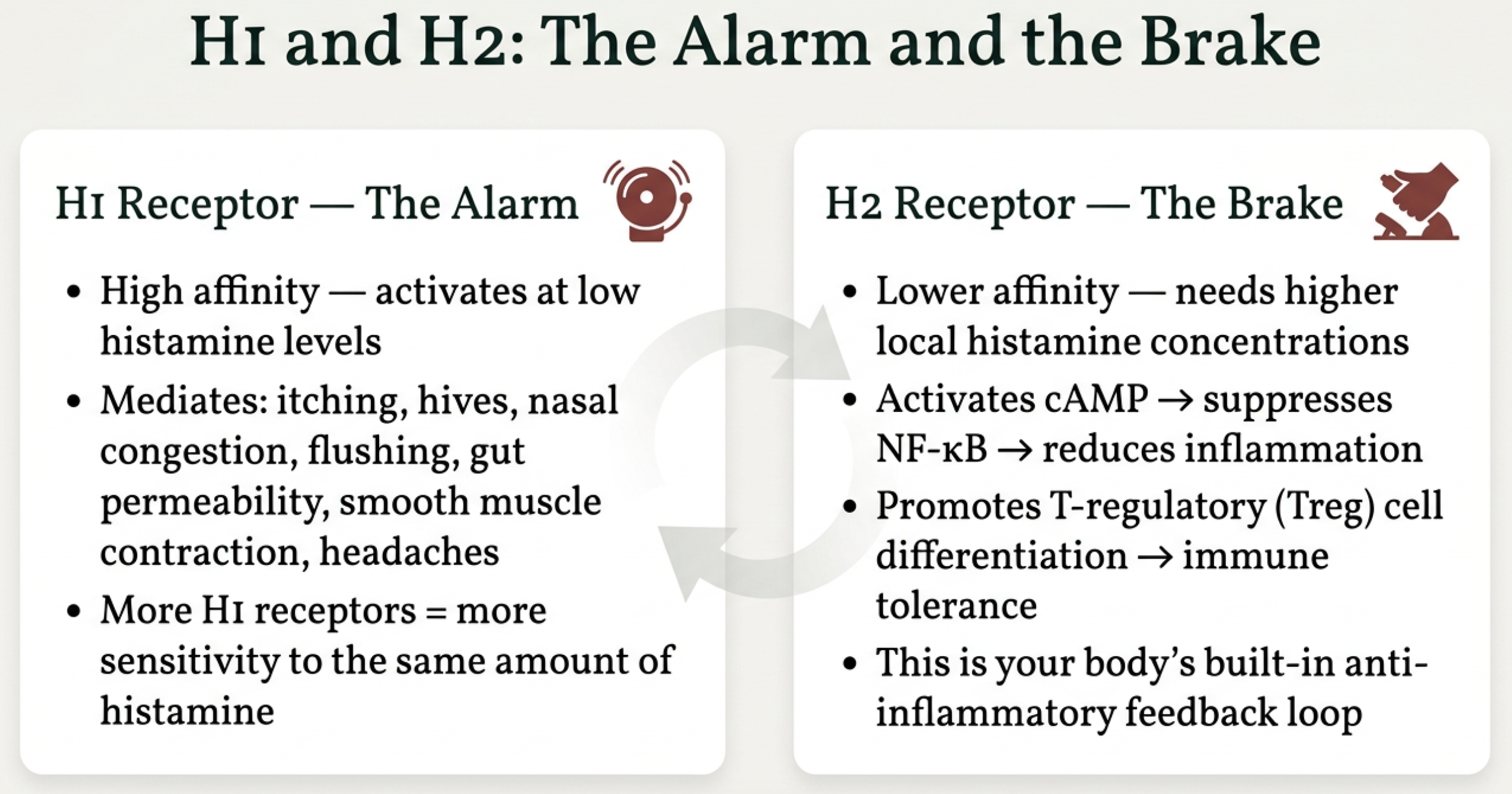
Two receptors matter most. The H1 receptor is the alarm — it's high-affinity, meaning it activates at low histamine levels, and it mediates the symptoms most people associate with histamine reactions: itching, hives, flushing, nasal congestion, headaches, and increased gut permeability [2]. The critical concept here is that the number of H1 receptors on your cells determines your sensitivity. More receptors means more reactivity to the same amount of histamine.
The H2 receptor is the brake. When activated, it triggers an anti-inflammatory cascade — increasing cAMP, suppressing NF-κB, and promoting T-regulatory cells [2]. It's your body's built-in feedback loop for calming the immune response.
What clears histamine — and this is where it gets important
Your body has two clearance enzymes, and they work in completely different compartments. Almost nobody talks about this distinction, and it changes everything about how you understand your symptoms.
DAO (diamine oxidase, encoded by the AOC1 gene) is your gut clearance enzyme. It's produced by enterocytes in the intestinal villi, and it stays local — degrading dietary histamine in the gut lumen before it can enter the bloodstream [3, 5]. DAO never reaches systemic circulation in meaningful quantities. It needs copper, zinc, vitamin B6 (as P5P), and vitamin C as cofactors, and alcohol is a direct competitive inhibitor [3].
HNMT (histamine N-methyltransferase) is your systemic clearance enzyme. It works inside cells — in the liver, kidneys, brain, and airways — and it's your only pathway for clearing histamine from mast cell degranulation, histamine in your skin, histamine in your brain, histamine in your vasculature [2, 3]. HNMT requires SAMe as its methyl donor, which is produced by the MAT enzyme, which requires magnesium. About 15–25% of the population carries a genetic variant (HNMT C314T) that reduces this enzyme's function [3].
How Does Dietary Histamine Actually Get Into Your System?
This is a question most HIT resources skip over, and the answer matters more than you might think.
Histamine is a small, positively charged molecule. That charge means it doesn't passively diffuse across intact cell membranes easily. It has two routes across the gut barrier.
The first route is through cells — organic cation transporters (OCT2, OCT3) move histamine into enterocytes, but once inside the cell, it immediately encounters DAO. Most histamine entering this way gets degraded before reaching the other side. There's a built-in metabolic checkpoint.
The second route is between cells — through tight junctions. When tight junctions are intact, this route is nearly closed. But when they open — via zonulin triggered by gluten, tryptase from mast cell degranulation, inflammatory cytokines, or nickel-driven mucosal inflammation — histamine flows through the gaps with no DAO checkpoint at all. It's an unregulated, unmetabolized leak directly into the bloodstream.
The bottom line: in a healthy gut with intact barrier function and normal DAO activity, dietary histamine is almost entirely neutralized before it reaches the systemic circulation. The diet only becomes a significant systemic contributor when the barrier is already broken.
The Honest Ranking: Where Is Your Systemic Histamine Actually Coming From?
This is what most HIT content creators don't tell you. The three sources of systemic histamine are not equal, and the one the low-histamine diet addresses is the smallest.
Number one: mast cell degranulation. This is by far the largest contributor to systemic histamine. Mast cells are distributed throughout every tissue — skin, gut mucosa, airways, around blood vessels [12]. When they degranulate, histamine is released directly into tissues and vasculature, completely bypassing DAO. DAO only operates in the gut lumen. A mast cell firing in your skin has zero DAO protection. Triggers include nickel via TLR4 [1], stress via CRH, allergens, infections, and temperature changes.
Number two: bacterial histamine production. HDC-positive bacteria living in or near the mucosa produce histamine continuously — not just at mealtimes [3, 5]. In SIBO or dysbiosis, they can locally saturate DAO even when the enzyme is functioning normally. Other bacterial metabolic byproducts like putrescine and cadaverine compete with histamine for DAO's active site, effectively crowding it out [3].
Number three: dietary histamine. Meal-dependent, episodic, and fully subject to the DAO barrier. Healthy people eat aged cheese, drink wine, and consume fermented foods every day without symptoms. Even scombroid poisoning — the extreme proof of concept — requires massive histamine levels exceeding 200 mg/kg to overwhelm normal DAO in a healthy person [3, 11].
I want to be transparent about the evidence basis here: this ranking is based on physiological reasoning and clinical pharmacology, not a controlled human study with compartmental modeling. The relative contributions have not been formally measured. But the logic is well-supported by what we understand about histamine pharmacokinetics, mast cell biology, and barrier physiology.
Your Symptoms Tell You Which Source Is the Problem
Here's where it gets clinically useful. The symptoms you get from local gut histamine versus systemic histamine are different — and the pattern is a diagnostic clue.
Local gut histamine — histamine acting directly on mucosal receptors — produces GI-dominant symptoms: bloating, diarrhea, cramping, nausea, acid reflux. These symptoms are dose-dependent, clearly linked to meals (20 minutes to a few hours), and they respond well to a low-histamine diet because you're reducing the load hitting those local receptors.
Systemic histamine — from mast cells firing throughout your body or from failed HNMT clearance — produces a completely different picture: flushing, hives, nasal congestion, headaches and migraines, brain fog, insomnia (because histamine is a CNS arousal neurotransmitter), rapid heartbeat, and dizziness [2, 8]. These symptoms are NOT tightly meal-linked and do NOT respond well to dietary restriction, because the primary source is mast cell degranulation, not food.
The diagnostic clue: if a low-histamine diet fixed your bloating and diarrhea but you still flush, still get migraines, still can't sleep, still have hives — that is the signature of unresolved systemic histamine. The diet addressed the local problem. The systemic source was never touched. This is the partial relief pattern.
The Missing Piece: Systemic Nickel Allergy Syndrome
An estimated 8–19% of the general population is sensitized to nickel [9, 10]. In sensitized individuals, dietary nickel triggers Systemic Nickel Allergy Syndrome (SNAS) — a condition that produces symptoms nearly identical to histamine intolerance: GI distress, skin flares, headaches, and brain fog.
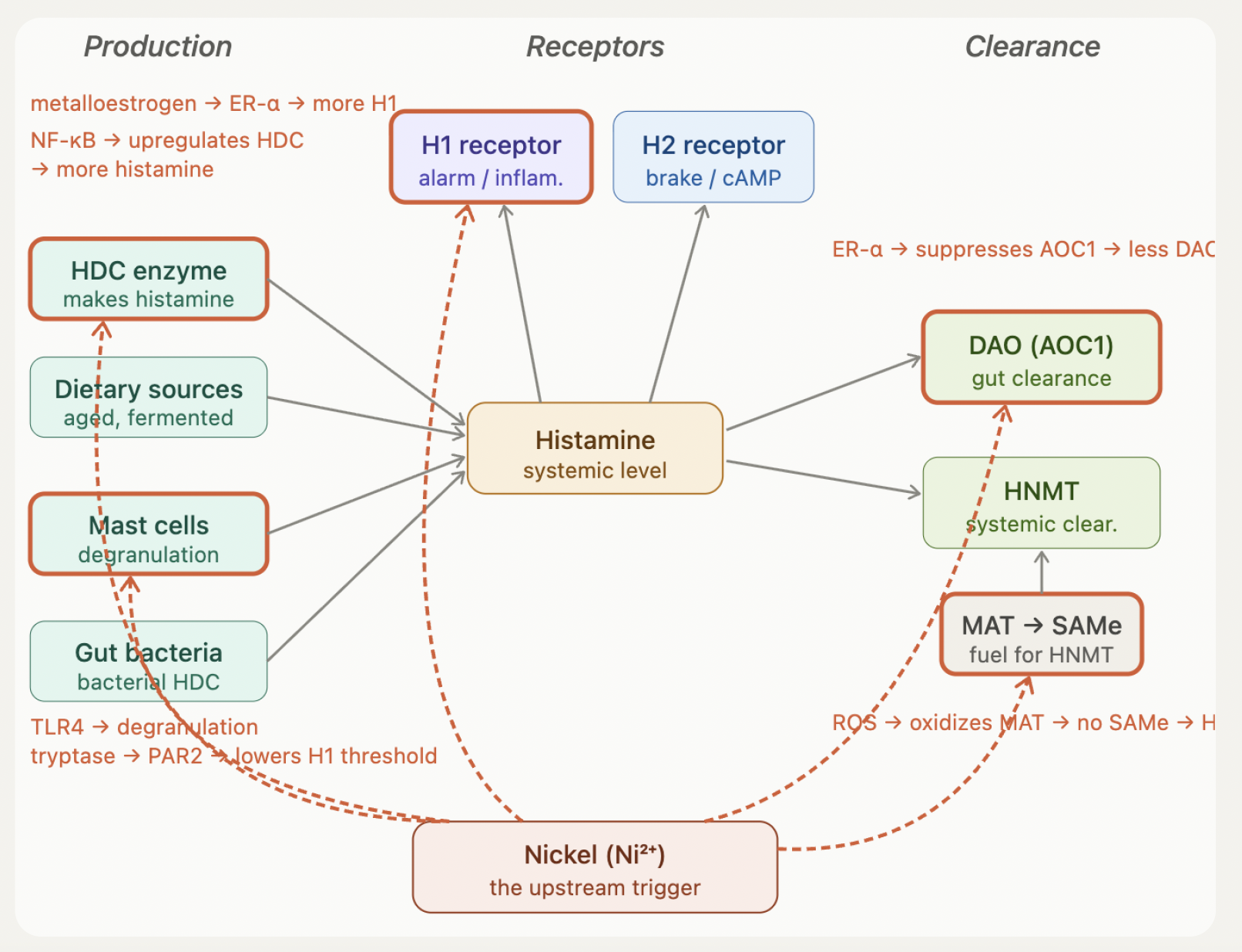
But nickel doesn't just mimic HIT. Research shows it attacks the histamine system at five distinct points.
Impact #1: Mast cell activation via TLR4
Nickel ions bind directly to the human TLR4 receptor, as demonstrated by Schmidt et al. in a landmark 2010 paper in Nature Immunology [1]. This triggers immediate mast cell degranulation — releasing stored histamine and tryptase. Tryptase activates PAR2 receptors, which lowers the activation threshold for H1. So you get more histamine released AND your alarm receptors become more sensitive. The delayed response includes NF-κB activation, driving TNF-α, IL-1β, and IL-6 — systemic inflammation from a dietary trigger.
Impact #2: H1 receptor upregulation
Nickel acts as a metalloestrogen — it mimics estrogen by binding to and activating estrogen receptor alpha (ER-α). ER-α activation upregulates the HRH1 gene, increasing the density of H1 receptors on your cells [2]. More receptors means greater sensitivity to the same amount of histamine. This is a key factor in why approximately 80% of HIT patients are middle-aged women — endogenous estrogen and nickel-mimicked estrogen are both pushing the same pathway.
Impact #3: DAO suppression
The same ER-α pathway that increases H1 receptors also downregulates AOC1 — the gene that encodes DAO [3]. Your gut's first-pass histamine defense is weakened from the inside by the same mechanism that's making you more sensitive.
Impact #4: HDC upregulation
NF-κB activation from TLR4 signaling upregulates the HDC gene — the enzyme that manufactures histamine from histidine [1, 2]. Your body now produces more histamine endogenously, independent of what you eat. This is why dietary restriction alone cannot solve the full problem.
Impact #5: MAT/SAMe/HNMT disruption
Nickel-induced inflammation generates reactive oxygen species (ROS). These ROS oxidize critical cysteine residues on the MAT enzyme, halting SAMe production [2]. Without SAMe, HNMT cannot methylate and inactivate histamine. Your systemic clearance — the only pathway that handles mast cell-derived histamine — collapses. If you also carry the HNMT C314T variant, this pathway was already compromised genetically. Nickel pushes it over the edge.
The complete picture
Nickel simultaneously increases histamine production (HDC upregulation + mast cell degranulation), increases sensitivity (H1 receptor density), and decreases both clearance pathways (DAO suppression + HNMT/MAT disruption). Every lever in the system — production up, sensitivity up, clearance down. The low-histamine diet touches one piece: dietary histamine in the gut lumen, which we've established is the smallest systemic contributor.
Why Cofactors Alone Aren't Enough
You may have heard the advice to supplement DAO cofactors — copper, zinc, B6, vitamin C — to support your enzymes. This is mechanistically correct, but it misses the rate-limiting step.
For DAO to work, four things must be true: the AOC1 gene must be transcribed (nickel suppresses this), mature enterocytes must exist at the villi tips (mucosal damage destroys them), the enzyme must not be competitively inhibited (alcohol and biogenic amines block it), and cofactors must be present. Most HIT patients are stuck at steps one through three. Supplementing step four when step one isn't happening is like putting premium gas in a car with no engine.
A specific caution on copper: true copper deficiency is rare outside of zinc oversupplementation, bariatric surgery, or prolonged TPN. Copper excess is more common and more dangerous — it's pro-oxidant and can worsen the very inflammatory environment that's already damaging MAT and the mucosa. Don't supplement copper without documenting a deficiency.
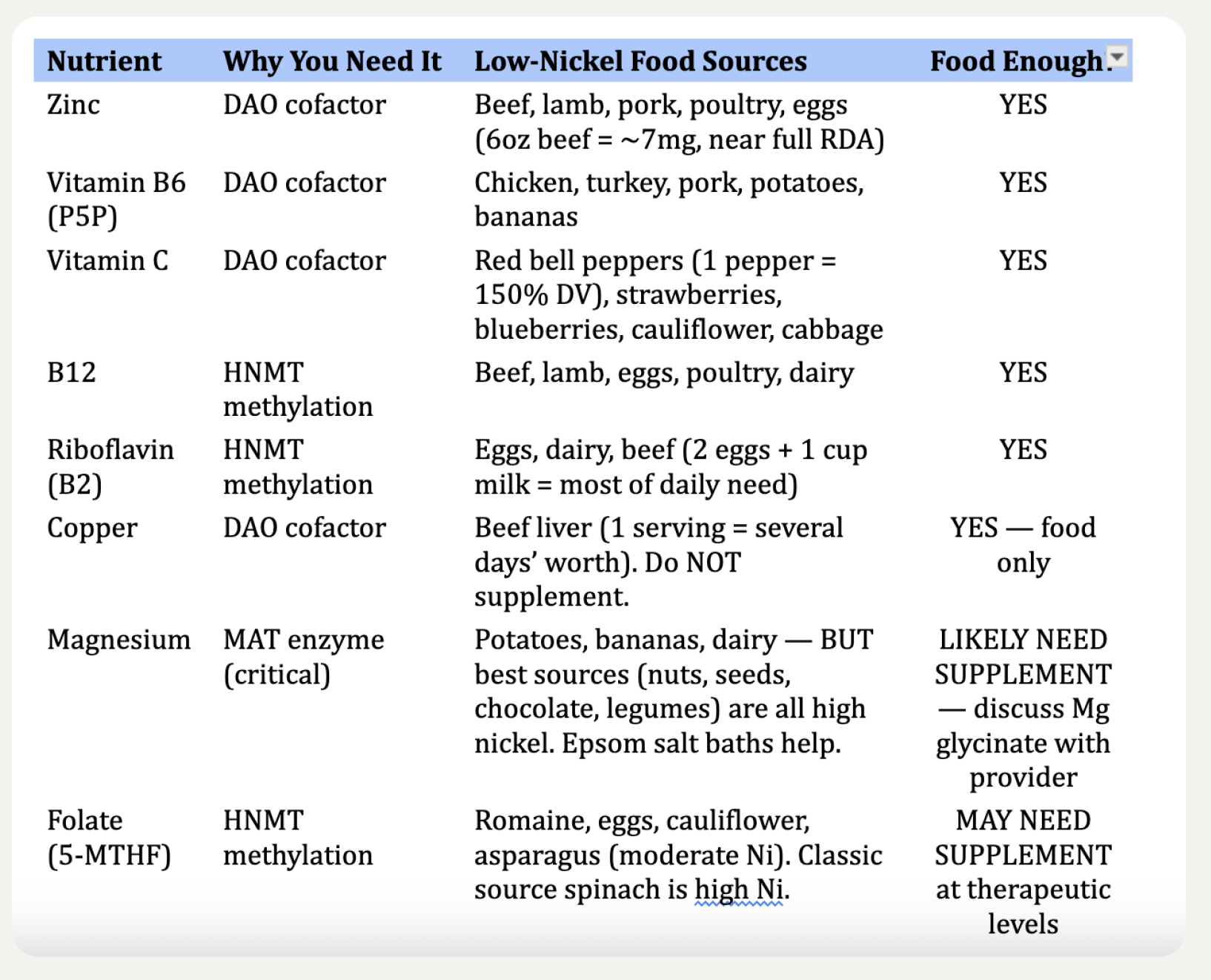
Food First: Getting Your Nutrients Without a Cabinet Full of Supplements
I know many of you are nervous about supplements — and for good reason. Nickel contamination in supplements is a real concern for SNAS patients. The good news is that you can get most of what your enzymes need from low-nickel foods.
Achievable through food alone:
Zinc (DAO cofactor) — beef, lamb, pork, poultry, eggs. A 6oz serving of beef provides roughly 7mg, close to the full RDA. Vitamin B6 (DAO cofactor) — chicken, turkey, pork, potatoes, bananas. Vitamin C (DAO cofactor) — red bell peppers (one medium pepper delivers 150% of your daily value), strawberries, blueberries, cauliflower, cabbage. B12 (HNMT methylation) — any animal protein. Riboflavin (HNMT methylation) — eggs, dairy, beef; two eggs plus a cup of milk covers most of the daily need. Copper (DAO cofactor) — beef liver is extraordinarily rich; one serving provides several days' worth. Do not supplement.
May need supplementation — discuss with your provider:
Magnesium is the hardest nutrient gap for SNAS patients. The best food sources — dark chocolate, nuts, seeds, legumes, whole grains — are all high in nickel. Low-nickel sources like potatoes, bananas, and dairy provide modest amounts but probably not enough to restore depleted MAT. Epsom salt baths (transdermal magnesium sulfate absorption) are a low-risk option. Magnesium glycinate supplementation is worth discussing with your provider because MAT literally cannot function without adequate magnesium.
Folate (5-MTHF) is also tricky — the classic source, spinach, is high in nickel. Romaine, eggs, and cauliflower provide some but likely not therapeutic levels.
The bottom line: two or three targeted supplements discussed with your provider — not a cabinet full of bottles. Food handles most of it.
The Pregnancy Proof: What It Actually Tells Us
During pregnancy, the placenta produces DAO at up to 500 times normal levels. Women with chronic migraines, urticaria, and systemic HIT symptoms often experience total remission in the second and third trimesters. Symptoms come right back after delivery [3].
Most people present this as evidence that "DAO support works." But they're missing the critical distinction between where that DAO is operating.
The DAO your intestines make stays in the gut lumen. It's local. It handles dietary histamine. It never reaches your bloodstream in meaningful quantities. This is also why oral DAO supplements produce modest, mostly GI-focused effects — they stay in the gut too.
Placental DAO is completely different. The placenta secretes DAO directly into the maternal bloodstream. At 500 times baseline, it acts as a massive circulating systemic histamine scavenger — mopping up histamine from mast cell degranulation, from failed HNMT clearance, from everywhere in the vasculature.
What pregnancy actually proves is that the bottleneck is systemic clearance, not dietary load. If food were the main problem, a low-histamine diet would have been sufficient — you wouldn't need a placenta flooding your bloodstream with 500x DAO to achieve full remission. In non-pregnant life, HNMT is your only systemic clearance pathway, and nickel is destroying it. Pregnancy provides a temporary bypass. Removing nickel provides a permanent fix.
The Integrated SNAS-HIT Protocol
Step 1 — Remove (stop the triggers): Implement a low nickel diet — the single intervention that addresses all five nickel impacts. Eliminate alcohol (direct DAO inhibitor). Reduce or eliminate gluten (triggers zonulin, opens tight junctions). Remove high-nickel prebiotics like inulin and FOS from chicory. Temporarily reduce high-histamine fermented foods until clearance capacity is restored.
Step 2 — Restore (rebuild the enzymes, after removing the trigger): For DAO: zinc, B6 as P5P, vitamin C from low-nickel sources like red bell peppers and blueberries. For MAT/HNMT: magnesium, methylcobalamin B12, 5-MTHF folate, riboflavin. TMG (betaine) to help clear SAH and prevent HNMT inhibition. Prioritize food sources first; supplement the genuine gaps (magnesium, possibly folate) with your provider's guidance.
Step 3 — Reinforce (repair the gut barrier): Butyrate-producing fibers like PHGG and acacia fiber. Retrograded starches from cooked-and-cooled white rice or potatoes. Tributyrin supplementation to directly upregulate occludin tight junction assembly. Zinc for tight junction integrity.
Step 4 — Modulate (calm the immune response): L. reuteri DSM 17938 provides anti-inflammatory benefits via adenosine/A2A receptor signaling — WITHOUT producing histamine. Unlike strain 6475 (clade II), DSM 17938 (clade VI) lacks the hdc gene cluster entirely, making it appropriate for histamine-intolerant patients. Many common strains — including *L. casei*, *L. bulgaricus*, and *L. reuteri* 6475 (clade II) — are histamine producers. Strain matters: only *L. reuteri* DSM 17938 (clade VI) is confirmed histamine-free.
What You Can Do Today
Cook fresh and eat fresh. If you meal prep, freeze portions immediately after cooking — don't let them sit in the fridge for days. Choose fresh meat over canned, cured, or processed. Replace high-nickel grains like oats and whole wheat with white rice and potatoes. Get your vitamin C from red bell peppers and blueberries, not supplements with unknown nickel contamination.
Prioritize sleep — histamine is an arousal neurotransmitter, and poor sleep increases H1 receptor sensitivity the next day [8]. Manage stress — CRH has direct receptors on mast cells, and stress-driven degranulation bypasses your diet entirely.
And if you've been supplementing B6 and zinc for months without improvement, consider the possibility that the upstream trigger was never addressed. The cofactors matter — but they only work once the engine is actually running.
Want the full breakdown with action checklists? Download the free Histamine Intolerance & SNAS Guide (click here).
My cookbook — The Low Nickel Diet Cookbook — features gut-health-promoting recipes designed specifically for the low nickel diet. Available at thelownickeldiet.com.
Low , Medium, and High Nickel Food Lists for free here
Join us for weekly live deep dives every Sunday at 11:00 AM MST on YouTube. Follow @thelownickeldiet everywhere.
Disclaimer: This post is for educational and informational purposes only and does not constitute medical advice. It is not intended to diagnose, treat, cure, or prevent any disease or condition. Individual responses to dietary changes and supplements vary. Always consult your physician, allergist, or qualified healthcare provider before making changes to your diet, supplement regimen, or treatment plan.
References
Schmidt M, Raghavan B, Müller V, et al. Crucial role for human Toll-like receptor 4 in the development of contact allergy to nickel. Nat Immunol. 2010;11(9):814-819. doi:10.1038/ni.1919
Jutel M, Akdis M, Akdis CA. Histamine, histamine receptors and their role in immune pathology. Clin Exp Allergy. 2009;39(12):1786-1800. doi:10.1111/j.1365-2222.2009.03374.x
Maintz L, Novak N. Histamine and histamine intolerance. Am J Clin Nutr. 2007;85(5):1185-1196. doi:10.1093/ajcn/85.5.1185
Comas-Basté O, Sánchez-Pérez S, Veciana-Nogués MT, Latorre-Moratalla ML, Vidal-Carou MC. Histamine intolerance: the current state of the art. Biomolecules. 2020;10(8):1181. doi:10.3390/biom10081181
Schnedl WJ, Enko D. Histamine intolerance originates in the gut. Nutrients. 2021;13(4):1262. doi:10.3390/nu13041262
Thomas CM, Hong T, van Pijkeren JP, et al. Histamine derived from probiotic Lactobacillus reuteri suppresses TNF via modulation of PKA and ERK signaling. PLoS One. 2012;7(2):e31951. doi:10.1371/journal.pone.0031951
Ma X, Fan PX, Li LS, Qiao SY, Zhang GL, Li DF. Butyrate promotes the recovering of intestinal wound healing through its positive effect on the tight junctions. J Anim Sci. 2012;90(Suppl 4):266-268. doi:10.2527/jas.50965
Krystal AD, Richelson E, Roth T. Review of the histamine system and the clinical effects of H1 antagonists: basis for a new model for understanding the effects of insomnia medications. Sleep Med Rev. 2013;17(4):263-272. doi:10.1016/j.smrv.2012.08.001
Ricciardi L, Arena A, Arena E, et al. Systemic nickel allergy syndrome. Ann Allergy Asthma Immunol. 2014;113(1):13-18.
Pizzutelli S. Systemic nickel hypersensitivity and diet: myth or reality? Eur Ann Allergy Clin Immunol. 2011;43(1):5-18.
Kovacova-Hanuskova E, Buday T, Gavliakova S, Plevkova J. Histamine, histamine intoxication and intolerance. Allergol Immunopathol (Madr). 2015;43(5):498-506. doi:10.1016/j.aller.2015.05.001
Bischoff SC. Physiological and pathophysiological functions of intestinal mast cells. Semin Immunopathol. 2009;31(2):159-177. doi:10.1007/s00281-009-0165-4